Recropping issues with pre emergent herbicides
Author: John Cameron & Mark Congreve (Independent Consultants Australia Network Pty Limited) | Date: 26 Feb 2016
Take home message
- The loss of many post-emergent herbicides to resistance is prompting increased use of pre-emergent herbicides
- With planning, pre-emergent herbicides can fit into parts of most farming systems
- Key breakdown mechanisms for most pre-emergent herbicides in the soil rely on one or both of microbial degradation or a chemical reaction known as hydrolysis. Both mechanisms need soil water in same zone as the herbicide
- Insufficient rainfall, or a rainfall pattern where very heavy falls are followed by prolonged dry spells, can significantly increase the duration of the required re-crop interval
- Herbicides broken down by hydrolysis include the triazines and sulfonylureas. Breakdown via hydrolysis is much slower at high pH levels - hence these products persist longer in alkaline soils, where microbial decomposition becomes the primary pathway for breakdown
- While the imidazolinones are broken down primarily by microbial activity, they bind far tighter to soil in low pH conditions, and are thus less available for microbial breakdown in low pH soils. As a result, these products persist longer in low pH soils
- For highly soluble, low binding herbicides, leaching may be a major pathway for herbicide loss. If there is an impervious layer in the soil profile, herbicide may sit above this layer and therefore persist longer than in a free draining soil
Weed resistance to post emergent herbicides is resulting in increasing use of pre-emergent herbicides. While solving a herbicide resistance issue with another herbicide is not a long term solution, pre-emergent herbicides can in the short term add much-needed diversity to the cropping system and provide a cost-effective weed control buffer while growers get their heads around the longer term need to integrate multiple non-herbicide tactics into their farming system.
Pre-emergent herbicides used in crop can be highly effective at protecting crop yield, but may allow sufficient escapes so that the weed seedbank is replenished. Escapes can occur due to uneven soil coverage and/or incorporation or weed seeds emerging from either above or below the herbicide band (depending upon the pre-emergent herbicide used). In the fallow, pre-emergent herbicides offer a much-needed option to reduce subsequent germinations, and work particularly well in already low density weed seedbank populations.
Typically, pre-emergent herbicides fit in fallow applications as a tank mix partner with paraquat in the second spray in a double knock sequence. By eliminating one or more subsequent weed germinations, there can be significant cost savings as well as benefits in spray logistics and management
More innovative uses for pre-emergent chemistries include targeting them for patch management and integrating their use with weed mapping software.
Many growers place great value in their ability to maintain cropping flexibility. As a result, post-emergent chemistries are often preferred. Growers that are unwilling to use pre-emergent herbicides forego the significant benefits and advantages of using these products in their farming system.
On any property in any season, there will almost always be some paddocks for which there is a known rotational plan where the downside risk of using pre-emergent herbicides on re-crop flexibility can be managed. Benefits of pre-emergent herbicides include:
- diversity of weed management tactics and mode of actions used
- reduced logistical pressure on post-emergent spraying
- capture of potential yield benefits from combining pre-emergent herbicides, early sowing opportunities and the high level of crop competition often associated with earlier sown crops.
With increased use of pre-emergent herbicides and recent drought conditions in many areas, a number of growers have been caught with herbicide residuals that impeded cropping flexibility. This paper focuses on the breakdown mechanisms of pre-emergent herbicides, after they have been incorporated either by rainfall or other means, and the implications this has for re-crop intervals.
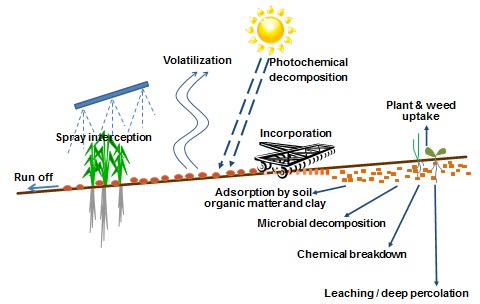
Figure 1. Pathways for herbicide degradation, loss and movement
Most pre-emergent herbicides are broken down by microbial activity in the soil. A smaller number are broken down primarily by a chemical reaction known as hydrolysis.
The rate of herbicide persistence is usually reported as a DT50 value. DT50 values represent the half-life, or the days for 50% of the herbicide in the soil to breakdown. As this varies for different soils and conditions, the DT50 is often reported as a range of values from different soil types or as an average value across a range of soils.
Herbicides with a DT50 under 30 are often classified as non-persistent as they tend to breakdown relatively quickly. However, at very high rates, some products with low DT50 values can still provide useful pre-emergent activity. An example is s-metolachlor e.g. Dual® Gold (DT50 of approximately 21). In the case of Dual Gold the use rate in sorghum and maize is 1-2L/ha (960-1920 gai/ha) which is much higher than required for short term residual weed control, however by applying this high rate, a few months residual control can be achieved before the product dissipates. The speed of breakdown in the soil depends on a range of factors, including:
- For products that are broken down by microbial processes:
- soil organic matter to support the microbial population in that zone of the soil where the herbicide residue lies
- supply of oxygen, nutrients and a relatively neutral pH
- warm/hot temperature will increase the speed of build-up of microbial populations
- rainfall pattern
- rainfall is generally the factor limiting the build-up of microbial populations, and hence herbicide breakdown
- populations will build fast following a rainfall event, while also crashing fast as the soil dries out, particularly in the soil surface where most of the microbes reside
- rainfall patterns which keep the soil wet for a number of weeks will see much faster degradation than patterns of high intensity rainfall followed by prolonged periods of dry soil conditions
- extended dry periods can significantly increase the expected half-life of herbicides in the soil
- microbial populations are lower at depth, where access to organic matter, nutrients, temperature and moisture are often lower. For mobile herbicides that move down the soil profile, the speed of microbial decomposition may be slower at depth, which can result in satisfactory establishment of a following crop, however symptoms may develop when the roots get down to residual herbicide layer.
- The amount of herbicide applied, i.e. a higher application rate will take longer to break down to a ‘safe’ level
- Herbicide binding characteristics
- herbicides that bind tightly to the organic matter and soil are often slower to breakdown via either microbial or chemical degradation. A herbicides strength of binding to clay or organic matter is reflected by the binding equilibrium coefficient (Koc) for that herbicide. The Koc is the ratio of herbicide adsorbed onto the organic matter and soil, compared to the amount present in soil water. A product with a very high Koc is very tightly bound, while a product with a low Koc is more available in the soil moisture and hence more available for plant uptake, movement and exposure to degradation processes.
- soil pH affects the imidazolinone herbicides, which bind far more strongly at lower pH and are thus less available for microbial degradation, so they persist longer at low pH, than at neutral or alkaline pH levels.
- Herbicide solubility
- products that are highly water soluble will be easily incorporated by rainfall and will move with the soil moisture.
- highly soluble herbicides are often more easily taken up by the plant, and hence may be more prone to causing crop injury after application
- herbicides with higher solubility and low binding often leach through the profile and this can often be a major pathway for herbicide loss.
- products that are highly water soluble will be easily incorporated by rainfall and will move with the soil moisture.
Table 1. Average adsorption coefficients for selected pre-emergent herbicides*
|
Herbicide (example) |
Average Koc value |
||
|
pendimethalin (Stomp®) |
17 581 |
Koc > 4 000 Non-mobile. |
Likely to bind tightly to soil and organic matter |
|
trifluralin (Treflan®) |
15 800 |
||
|
tri-allate (Avadex®) |
3 034 |
Koc 500 to 4 000 Slightly mobile |
|
|
diflufenican (Brodal®) |
1622 - 2369 |
||
|
prosulfocarb (Boxer® Goldp) |
1367 - 2340 |
||
|
diuron (various) |
813 |
||
|
terbuthylazine (Terbyne®) |
230 |
Koc 75 to 500 Moderately mobile |
Likely to move with soil water |
|
dimethenamid-P (Outlook®) |
218 |
||
|
s-metolachlor (Dual®) |
200 |
||
|
isoxaflutole (Balance®) |
145 |
||
|
imazapic (Flame®) |
137 |
||
|
simazine (Gesatop®) |
130 |
||
|
atrazine (Gesaprim®) |
100 |
||
|
pyroxasulfone (Sakura®) |
95 |
||
|
metribuzin (Sencor®) |
60 |
Koc 15 to 75 Mobile |
|
|
triasulfuron (Logran®) |
60 |
||
|
chlorsulfuron (Glean®) |
40 |
||
|
picloram (Tordon®) |
13 |
Koc 0 to 15 Very mobile |
|
|
clopyralid (Lontrel®) |
5 |
||
p Boxer Gold also contains s-metolachlor
*Source: The soil behaviour of pre-emergent herbicides – a manual for Australian grains advisers, GRDC 2014
Table 2. Primary breakdown mechanisms and speed of breakdown for selected herbicides as represented by the DT50.
|
Herbicide (example) |
Ave DT50 values (range) |
Primary breakdown pathway(s) in the soil |
|
isoxaflutole (Balance®) |
1.3 |
Chemical hydrolysis and microbial degradation |
|
dimethenamid-P (Outlook®) |
7 (2-16) |
Microbial |
|
prosulfocarb (Boxer® Goldp) |
10 (7-13) |
Microbial |
|
metribuzin (Sencor®) |
14-28 |
Microbial |
|
triasulfuron (Logran®) |
19 (3-48) |
Hydrolysis is the primary pathway in neutral and acidic soils. Slow microbial degradation is the primary pathway in alkaline soils |
|
s-metolachlor (Dual®) |
21 (11-31) |
Volatility generally low but photo degradation can be high prior to incorporation. Microbial degradation |
|
terbuthylazine (Terbyne®) |
22 (10-36) |
Hydrolysis is the primary pathway in neutral and acidic soils. Slow microbial degradation is the primary pathway in alkaline soils. |
|
pyroxasulfone (Sakura®) |
22 (16-26) |
Microbial via cleavage of the methyl-sulfone bridge |
|
chlorsulfuron (Glean®) |
36 (11-70) |
Hydrolysis is the primary pathway in neutral and acidic soils. Slow microbial degradation and slow hydrolysis are the primary pathways in alkaline soils |
|
clopyralid (Lontrel®) |
40 (12-70) |
Leaching can be significant. Microbial degradation, fastest in warm, moist soils |
|
tri-allate (Avadex®) |
46 (8-205) |
Volatilisation and microbial |
|
atrazine (Gesaprim®) |
60 (6-108) |
Hydrolysis is the primary pathway in neutral and acidic soils. Slow microbial degradation is the primary pathway in alkaline soils. |
|
diuron (various) |
89 (20-231) |
Volatilisation and photo degradation losses are typically low, but can be significant under hot and dry conditions if not incorporated. Slow microbial degradation in the soil. |
|
simazine (Gesatop®) |
90 |
Hydrolysis is the primary pathway in neutral and acidic soils. Slow microbial degradation is the primary pathway in alkaline soils. |
|
picloram (Tordon®) |
90 (20-300) |
Photolysis and leaching can be significant. Slow microbial degradation. Fastest under warm moist conditions. |
|
pendimethalin (Stomp®) |
90 (27-186) |
Subject to photo degradation and volatility loss if not incorporated. Slow microbial degradation in the soil, rapid under waterlogged conditions. |
|
trifluralin (Treflan®) |
170 (35-375) |
|
|
diflufenican (Brodal®) |
180 |
Binds tightly to soil. Metabolic degradation is slow. |
|
imazapic (Flame®) |
232 (31-410) |
Slow microbial degradation. Binding increases in acidic soils which increases persistence. |
DT50 0-30 = relatively non persistent, DT50 30-100 = moderately persistent, DT50 >100 = persistent
p Boxer Gold also contains s-metolachlor
*Source: The soil behaviour of pre-emergent herbicides – a manual for Australian grains advisers, GRDC 2014
As already stated, safe re-cropping intervals are often more reliant on the period of time that soil is wet for (to sustain a microbial population in the case of herbicides broken down by microbes, or to support the chemical process for herbicides broken down by hydrolysis), rather than the total amount of rainfall received during the re-crop interval.
Recommendations on product labels reflect expected safe re-crop intervals based on trials conducted over a range of climatic experiences. It is however entirely unrealistic to expect that any set of field trials, no matter how extensive, will reflect the full range of soil and environmental conditions that can be experienced over time and across geographies.
The New South Wales DPI publications ‘Weed control in winter crops’ and ‘Weed control in summer crops’ provide detailed plant back tables for most herbicides used in New South Wales. Product labels are the primary source of information and should be read prior to herbicide use.
In situations where growers are concerned that soil residuals may still be present, the safest option is to plant a crop or varietal type with tolerance to the herbicide in question.
Soil tests are rarely used as they are highly specialist in nature; there is limited experience to help interpret test results if some herbicide is detected and they are costly to undertake. If soil tests are to be used, then it is also essential to consider where the herbicide is expected to be in the profile to ensure that soil samples are taken from the right depths. For example, if only the 0-5cm zone is tested and was to reveal no herbicide residue it is still possible to have herbicide residues remaining at depth which can be picked up by the roots of a subsequent crop later in the season.
Paddock bioassays with ‘canary’ species (a canary species is a highly sensitive species to that herbicide), can provide some insight, however they can give inaccurate feedback on the likely level of crop safety. An example here would be with the imidazolinones or sulphonylureas. These products at sub lethal dose rates do not stop emergence from occurring. Affected plants often emerge and appear as if they have escaped the herbicide effect, only to later show significant suppression and stunting (often called the green skeleton effect). Without a valid comparison and the ability to allow plants to grow for an extended period of time, such tests can give a false sense of security and should be used with caution.
For herbicides that are tightly bound to the soil surface, aggressive cultivation prior to sowing is recommended on the label of some herbicides. This may dilute remaining herbicide in the soil profile, resulting in improved crop establishment. This process is not without risk.
Where herbicide residue may remain in the soil, avoid the use of herbicides from the same mode of action group in following crops. It is not uncommon to see a herbicide stress acting on top of an existing herbicide stress to make a potentially damaging residual situation worse. An example of this would be a crop of a non-Clearfield variety of wheat sown into a paddock where an inadequate level of rainfall had occurred to breakdown an imidazolinone residue. Imidazolinones are group B (ALS inhibitor) herbicides. If a another group B ALS inhibitor herbicide such as a sulphonylurea was applied to a wheat crop that was already affected by and trying to cope with an imidazolinone residual this could be enough to tip it over the edge and result in significant crop damage. The ability of the wheat crop to survive sulphonylurea herbicides is based on its ability to detoxify the herbicide via metabolic processes. If crop metabolism is already slowed as it is dealing with a dose of another herbicide from the same herbicide mode of action group this could turn a low-level damage incident into a far higher one.
Good agronomic practice that promotes early crop health and vigour can assist in overcoming some low-level marginal damage. While any level of herbicide damage or setback to a young crop may potentially lead to a yield loss or change in phenology, and should thus be avoided, it should be noted that it is not uncommon for crops suffering from low-level herbicide damage in the early vegetative phases of growth, to compensate and yield well despite their early setback. Growers relying on the crops ability to compensate and grow out of early damage are however taking a significant risk.
Further reading
The soil behaviour of pre-emergent herbicides - a reference manual for Australian grains advisers. Grains Research and Development Corporation, 2014.
Acknowledgements
The research undertaken as part of this project is made possible by the significant contributions of growers through the support of the GRDC, the author would like to thank the GRDC for their continued support
Contact details
John Cameron
Independent Consultants Australia Network Pty. Ltd.
PO BOX 718, Hornsby NSW 1630
Ph: 02 9482 4930
Email: john@icanrural.com.au
® Registered trademark
GRDC Project Code: ICN00016,
Was this page helpful?
YOUR FEEDBACK
