New insights into slug and snail control
Abstract
Snail and slug baits should be considered as crop protectants. Cultural methods are required to reduce populations and the biological function of farming systems needs to be considered. Baits often perform badly and have to be re-applied due to field degradation and/or pest populations not actively feeding. Research has focused on improving bait performance.
Baits have a limited field life, with the commonly used bran based metaldehyde products (e.g. Meta®) lasting less than two weeks, thus need to be applied regularly. Rainfall not only physically breaks down bran pellets; it causes a reduction in number of snails killed. Temperature, not UV light, also reduces the efficacy of metaldehyde baits. Choose the product based on cost and the context in which it will be applied. Different products perform differently. Rules of thumb: do not use current iron based baits when >10mm rain is expected; do not use Metaldehyde products over the summer and expect them to last.
Observations using time lapse cameras and recording environmental factors highlight different species are active and feed at different times. Even within populations, individuals behave differently depending on the moisture conditions where they hang out. Weather data from the Bureau of Meteorology (BoM) is not as accurate as ground moisture, or soil moisture in predicting behaviour. Using detailed micro-climate information and individual snail’s behaviour to inform bait application and timing of cultural controls resulted in good field control in 2015.
The role of crop rotations needs to be considered; canola leads to a build-up in numbers and linseed has been found to limit slug populations. How will early sown canola influence populations?
Snails can be controlled in no-till, full stubble systems once growers understand the context of where and when controls are applied, and follow a few basic guidelines. Bait needs to be applied when snails are active and feeding, with the timing varying depending on paddock and seasonal conditions and the species present. Basic rule of thumb: bait snails in the autumn when snails are actively feeding and prior to egg laying.
Background
Several exotic snail and slug species of European-Mediterranean origin have established in Australia and become important pests of grain crops. Snails cause substantial economic losses through yield loss from feeding damage, field control costs, additional harvest costs, grain value loss, receival rejection and threaten market access.
The biggest threat from snails is market access, with common white or vineyard snail (Cernuella virgata) now listed in formal import standards (February 2015) for wheat and barley into China. The Chinese market is worth $1.5 billion (2014 ABARE) to Australia. Previous contamination issues (e.g. Korea 2012) highlight the potential cost snails pose to the grains industry, when a major market restricts access due to a quarantine breach.
Crop losses are difficult to quantify. Estimates have indicated an overall enterprise cost to farms with snails is $33/ha, with $27/ha spent on bait and $17/t spent on post-harvest cleaning (GRDC 2015 report). For individual farm enterprises, snail costs were equivalent to 14 per cent of the farm profit in the Yorke Peninsula/Lower North region of South Australia (SA) and 34 per cent and 44 per cent of farm profits in the less profitable Victorian Wimmera and Mallee, respectively (2000 report).
Where snails and slugs are a high risk (south east SA and western Victoria (Vic)) growers have shifted from growing canola and/or have implemented strategic burning and cultivation. That opportunity cost is estimated upwards of $270 million annually lost to the canola industry alone. A five per cent production loss by slug and snail activity would represent >$82 million loss to the Australian canola industry (2012 values).
Research has indicated bait timing is an essential factor to limit snail contamination. Growers on Yorke Peninsula have responded by now baiting in March rather than after sowing in May/ June. In all operations, baiting is just one part of the year-round control program with cultural methods vital (e.g. cabling and rolling).
The overall aim of research presented in this paper is to improve decisions on bait applications due to the limited field life of current products and the variable feeding of the targets; that is snails and slugs do not always feed on baits. The first aim is to understand factors that lead to the degradation of bait products and compare the field life of various products. The second aim is to understand activity and feeding triggers, and what seasonal factors lead to greater pest numbers.
Methodology
Bait degradation
A number of experiments were conducted to give a range of conditions and test the effect of exposure time, and subsequently tease apart individual factors; temperature, ultra-violet (UV) light, rainfall and mould. Bait products were weathered by spreading approximately 50g of each on the surface of soil (Warooka red loam) in large planter trays (400 x 300 x 120 mm). Trays were placed on benches in an exposed position at the Waite Campus, Urrbrae, SA. Baits were exposed to weathering for seven periods during 2014, at seven day intervals (0, 7, 14, 21, 28, 35) resulting in a variety of conditions. Rain fastness of 16 different products was assessed twice by exposing to rainfall (>35 mm) over 14 days in 2015.
Italian snails (Theba pisana) were used to test the efficacy of molluscicidal baits once they had been exposed to the environment on soil. Five snails, eight replicates per treatment (n=40) were added to each test arena with eight baits as soon as practicable following the completion of weathering periods (usually within one week). Baits were removed three days after initiation of the experiment due to the formation of mould forming, which was scored as present/absent and the number and condition of pellets remaining recorded. Snail mortality was assessed five days after bait was removed.
Snail and slug activity
Ten paddocks across southern Australia have been intensively monitored using cameras to capture slug and snail activity (The blueprint on how to conduct remote sensing of snails - A Snug Blog). Environmental data was also collected: soil moisture and temperature (10cm); ground leaf wetness, temperature and relative humidity. Rainfall and barometric data was obtained from BoM. Snails were collected monthly (more frequently during autumn) for assessment of size (n=90), moisture content (n=45) and reproductive stage (n=45) achieved using digital calipers for shell diameter, before and after oven-drying weights (40°C for > four weeks), and albumen gland dissections, respectively.
Results and discussion
How will the latest research findings affect management strategies and packages?
Research findings regarding the field degradation of baits are presented to inform management about their likely efficacy under various weather conditions. Manufacturers often make claims about rain fastness; however these are based on physical integrity that was found not to influence actual efficacy (Table 1). Conclusions from this work:
- Rainfall erodes physical integrity of bran-based baits.
- Mould on products did not influence bait consumption or efficacy.
- Reduction of active ingredients by rainfall (metaldehyde & iron chelate) is most important.
- Individuals are more likely to consume a sub lethal dose.
- Don’t use current iron based baits when >10mm rain is expected.
- Temperature, not UV light, degrades metaldehyde baits.
- Don’t use metaldehyde products over the summer and expect them to last > 2 weeks.
- Commonly used bran products need to be re-applied < 2 weeks, more expensive products will last 3-4 weeks.
- Work out the cost benefit yourselves!
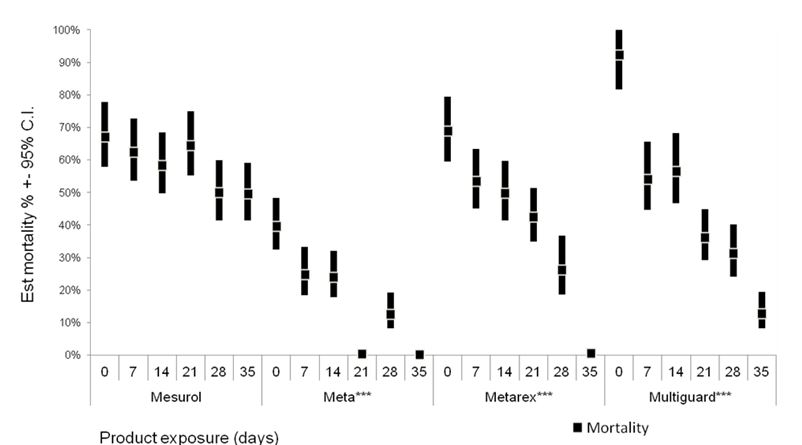
Figure 1: Product degradation (as reflected by snail mortality) in response to exposure to various weather conditions in 2014.
Overall estimates from seven experiments are presented as mean mortality and 95% confidence intervals, calculated from estimating number of dead snails using a generalized linear model (GLM) (Poisson, log link). Significant effect of exposure time is indicated by *** (P < 0.001).
Table 1: Response of various products to rainfall (>35mm) over 2 week period in 2015.
| Product |
Active ingredient | Conc. | Different groups no rain | Mortality |
Rainfall Sig. Dif. | Rank after Rainfall | |
|---|---|---|---|---|---|---|---|
| 2 weeks | 35 mm | ||||||
| Multiguard | Iron | 6% | a | 97% | 0% | Y | 12 |
| Mesurol | Methiocarb | 2% | a | 94% | 56% | N | 1 |
| Eradicate | Iron | 6% | a | 90% | 0% | Y | 12 |
| Ex 5 | Methiocarb | 2% | ab | 80% | 43% | Y | 2 |
| Ex 1 | Metaldehyde | 3% | bc | 59% | 21% | Y | 6 |
| Ex 3 | Iron | NS | cd | 53% | 0% | Y | 12 |
| Metakill (green) | Metaldehyde | 5% | cd | 51% | 24% | Y | 4 |
| Metarex micro | Metaldehyde | 5% | cd | 51% | 23% | Y | 5 |
| Ex 2 | Metaldehyde+ | 1.8% | cde | 49% | 6% | Y | 9 |
| Ex 4 | Metaldehyde+ | 5% | cdef | 44% | 23% | N | 5 |
| Metarex | Metaldehyde | 5% | cdef | 43% | 39% | N | 3 |
| Slugout | Metaldehyde | 1.8% | defgh | 29% | 16% | N | 7 |
| Metakill (blue) | Metaldehyde | 5% | efghi | 24% | 6% | N | 9 |
| Meta | Metaldehyde | 1.5% | ghi | 17% | 7% | N | 8 |
| Sluggoff | Metaldehyde | 3% | hi | 11% | 4% | N | 10 |
| Slugger 2.5mm | Metaldehyde | 1.5% | hi | 7% | 1% | N | 11 |
| Placebo | Nil | 0% | i | 0% | NA | NA | NA |
Combined mortality data (%) from the two experiments (14 reps per group) are presented with significant differences between ‘exposed to rain’ and ‘exposed to dry’ two-week treatments for a single product indicated by different letters. Rainfall resulted in a significant reduction (Χ216 = 658, P <0.001) in efficacy that interacted with product, hence significant differences between individual products is indicated with “Y” (HSD < 0.05), but due to variability we were not able to detect significance when less than a 25 per cent effect size.
Snail activity
Observations, including video footage, have led to more questions than answers. Wetness at 10cm above the ground seems to be best associated with round snail activity, although BoM relative humidity can be used; in summer snail activity triggered by 90 per cent relative humidity or higher. By late March this response is at 80 per cent relative humidity coinciding with the commencement of mating. Common white snails move more during the night, whereas small pointed snails often move during daylight in the early morning. During wet conditions pointed (conical) snails will move at similar times to round snails. It seems pointed snails (both species) need more moisture to become active, such as longer periods of high humidity or light showers. Observed species differences could be due to temperature, but also behavioural differences. For example, pointed snails have a staggered activity; some are active early in the season before the majority become active mid to late season. One theory is the staggered activity is similar to germination of weeds where buried seeds germinate at a different time to those on the surface. That is, pointed snails in the soil become active at a different time to those hiding up on stubble. This research highlights different species behave differently - bait application needs to match feeding activity.
Snail research
The other observation is, snails often move straight past a bait pellet, making us question claims about pellet attractiveness. Is their response to bait related to their physiological state? That is, does body moisture content of the snail or reproductive stage, as measured by albumen gland size, influence feeding on baits, hence ingestion of a lethal dose. Body moisture content of snails fluctuates during the year in response to available water in the environment. Albumen glands swell during reproductive activity. Enlarged glands indicate active egg-laying. Dissections of two populations of Italian white snails and three populations of common white snails within SA in 2015 indicated that glands began to swell in March 2015, peaking late April to mid-May. Common white snails at Palmer SA displayed a potential relationship between these characters (Figure 2), however a specific body moisture ‘trigger value’ for albumen gland enlargement and subsequent egg-laying is yet to be determined. Incorporating climate data will help predict when body moisture increases and combined with camera observations, the aim is to use site-specific weather data to help pin point optimal baiting periods. In 2015 information from a camera and weather station was used to inform bait application (10kg/ha Metaɸ) in mid-January when snail body moisture was >65% and prior to breeding as indicated by reduced size of albumen glands (Figure 2). This baiting followed rolling in early January, which resulted in >90% mortality. Despite no other bait applied for the season there was limited build-up of common white snails in that 2015 wheat crop.
ɸRegistered label rate is 5-7.5kg/ha
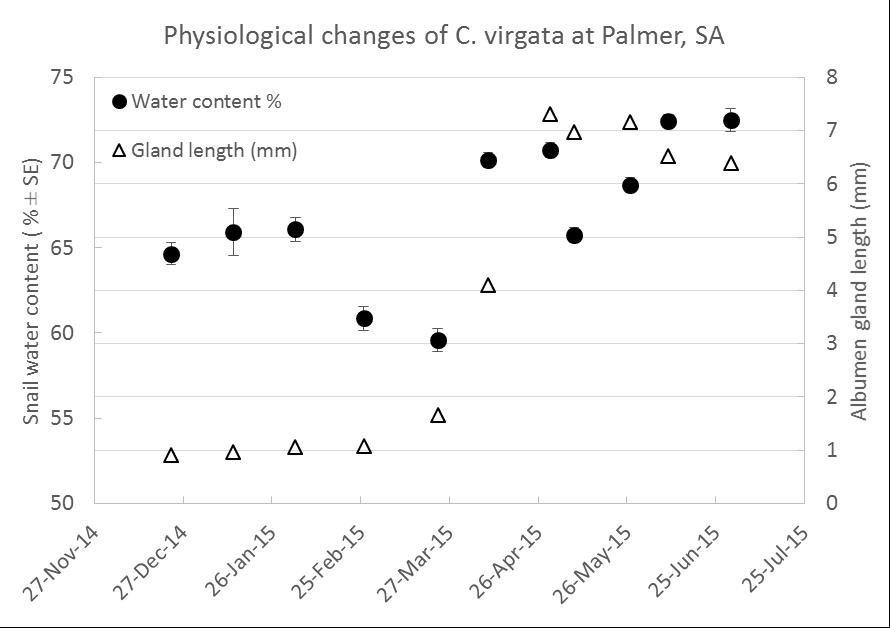
Figure 2: Changes in common white snail moisture and reproductive stage from summer to winter.
What are the risk factors that lead to increasing snail populations?
Improving soils and moisture holding capacity, which includes increasing macropores (porosity), organic matter and available calcium makes a more favourable habitat for snails and slugs. Moisture is the biggest determining factor for breeding, so in seasons that are more favourable for growing crops, populations will build up.
Table 2: Risk factors for snail outbreaks.
| Factor | High risk | Reduced risk | Low risk |
|---|---|---|---|
| Annual rainfall | Above average autumn and summer rainfall | Drought | |
| Stubble management | No till stubble retained | Tillage or burnt only | Tillage and burnt stubbles |
| Grazing livestock | No sheep in enterprise | Sheep on stubbles | |
| Soil | Alkaline calcareous soils | Un clayed non wetted sandy soils | acid soils with low organic matter |
| Weeds | Summer volunteers / Brassica weeds | No volunteers | |
| Previous paddock history of snails | No history of snails | ||
| Previous paddock history | Snails appear to build up most rapidly in canola, field peas and faba beans but can feed and multiply in all crops and pastures | Clean cereal crops | Poor cereal crop |
Other considerations
Baits should be considered as crop protectants. Cultural methods are required to reduce populations and the biological function of farming systems needs to be considered. Baiting for snails occurs at a different time to slugs. If you have both snails and slugs, one bait in April may not be good enough. Assess your risk and context in which you are trying to control these pests. Different products will suit different situations.
Consider crop rotation in high risk situations; bait snails in the summer/autumn when actively feeding which depends on moisture. Use cultural methods to control snails when hot. Consider what other pests are eating the snail bait. Metaldehyd baits are not reliable to control snails in crop as they do not work as well when cold.
Useful resources
Pestnotes on snail and slug biology at: A Snug Blog
Ground Cover TV episodes on slug monitoring and bait timing are available
Acknowledgements
We would like to thank John Midwood and Paul Breust, (SFS), Ken Young and Jen Lillecrapp (GRDC), Alan Mayfield (SAGIT) and Felicity Turner (MFMG) for comments and intellectual input.
Contact details
Helen DeGraafSARDI Entomology Unit, GPO Box 397, Adelaide SA 5001
08 8303 9543
helen.degraaf@sa.gov.au
Was this page helpful?
YOUR FEEDBACK
