Does combined use of organic and mineral phosphorus fertilisers support mycorrhizal colonisation
Take home messages
- Incorporation of organic amendments (OA) into phosphorus (P) management plans can have beneficial effects on arbuscular mycorrhizas (AM).
- OA alone may not be able to meet crop P demands.
- Combined use of OA and mineral P fertilisers successfully met crop P demands.
- Bicarbonate-extractable P gives a good indication of the P fertiliser potential of OA.
Background
Most phosphorus (P) fertilisers currently used come from phosphate rock, which is a finite resource (Scholz and Wellmer, 2013). Therefore, alternative sources of P fertilisers need to be used to ensure the sustainability of food production systems. Organic amendments (OA), such as manures and composts, can be used as P fertilisers. However, OA vary in the amount and forms of P that they contain (Sharpley and Moyer, 2000), which can affect their suitability as P fertilisers.Arbuscular mycorrhizas (AM) — symbioses between plant roots and fungi — can enhance plant P uptake (Pearson and Jakobsen, 1993). They also provide other benefits to soil and plant health (Gianinazzi et al., 2010). While AM are negatively affected by mineral P fertiliser addition (Abbott et al., 1984), the effect of OA on AM is not well understood. In this project, four OA were investigated for their potential to be used as P fertilisers. The relationship between the chemical properties of the OA and plant (wheat) P uptake from the OA was determined. A second experiment was conducted to determine whether chicken litter with straw bedding can be effectively used in combination with mineral P fertilisers, supplying crops with P while having minimal effect on AM colonisation. This work is part of a PhD funded by a GRDC Grains Research Scholarship.
Methods
Experiment One
Four OA were investigated — a pig litter with straw bedding, a chicken litter with straw bedding, a chicken litter with sawdust bedding and a municipal waste compost. These OA were analysed for their total P content (inductively coupled plasma-mass spectrometry (ICP-MS) following digestion in aqua regia), bicarbonate-extractable P content (following Colwell, 1963)), and P forms (using nuclear magnetic resonance (NMR) spectroscopy — for more details, see Mackay et al. 2017). Availability of P in the OA to plants was then determined in a pot experiment in which OA were applied to a low P, sandy loam soil at the same rate of total P – 20kg of P per ha. This equated to 1.2-8.0t of OA per ha. A treatment where phosphoric acid was applied at the same P rate was included as a completely inorganic control. A negative control was also included, in which no P was added to soil. The availability of the P in the amendments was then determined by growing wheat (var. Axe) from seed in the soil for 30 days and determining the P content of the plants.
Experiment Two
The same chicken litter with straw bedding and soil in Experiment One was used. Phosphoric acid and/or chicken litter were applied to soil at various rates and combinations. Wheat (var. Axe) was grown from seed in pots for 48 days and plant P content was determined. Roots were analysed for AM colonisation.
Results and discussion
Experiment One
Total P concentrations of the OA ranged from 2.5g/kg (compost) to 16.5 g/kg (chicken litter with straw bedding, Table 1). The percent of total P that was bicarbonate extractable was highest for the pig litter (59%) and lowest for the compost (24%, Table 1). Phosphorus forms, determined using NMR spectroscopy, could not be deconvoluted for compost samples. For the other OA, the percent of P which was orthophosphate ranged from 54% (chicken litter with sawdust bedding) to 77.3% (pig litter, Table 1). Other P forms which were present in high amounts included phytate and phospholipids (for more details, see Mackay et al., 2017). Based on these results, we predicted that if the OA were applied at the same rate of total P, the pig litter would provide the most P to plants and the compost would supply the least P to plants.
Table 1. Total P concentration of organic amendments used and the proportion of this P that is bicarbonate-extractable and orthophosphate. Adapted from Mackay et al. (2017).
| Chicken litter (straw) | Chicken litter (sawdust) | Pig litter (straw) | Compost | |
|---|---|---|---|---|
| Total P (g/kg) | 16.5 | 14.3 | 5.9 | 2.5 |
| Percent of P which is bicarbonate-extractable | 37.0 | 29.4 | 59.3 | 24.0 |
| Percent of P which is orthophosphate | 65.2 | 54.4 | 77.3 | n/a |
After 30 days of plant growth, plant P uptake varied considerably among treatments (Figure 1). Plants grown in the phosphoric acid treatment took up more than double the amount of P taken up by plants in OA treatments. All plants in OA treatments took up more P than the negative control, however, this was not significant for the compost treatment. Of the OA, the pig straw provided the most P to plants and the compost the least, as predicted. While the two chicken litters differed in their bicarbonate-extractable P and orthophosphate concentrations, they provided the same amount of P to plants. Despite this, these two P measurements gave a far better indication of P fertiliser potential compared to total P analysis. Given the cost difference between these two measurements, we recommend the bicarbonate-extractable P method to determine the P fertiliser potential of OA.
The OA investigated here would need to be applied at greater rates in order to provide plants with enough P to meet growth requirements, however, this may not be economically feasible. Moreover, while the P in the OA was not available to plants in this 30 day growth experiment, it may slowly become available to plants over longer time periods. Therefore, we predicted that OA, with their slow release of P, could be coupled with a mineral P fertiliser which is more readily available to crops, to supply crops with sufficient P while limiting excess P additions.
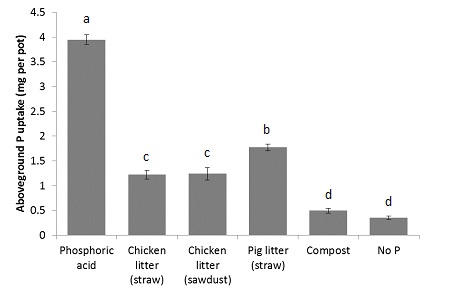
Figure 1. Above ground plant P uptake (content) per pot of plants grown in a greenhouse experiment where P was applied at the same rate as either phosphoric acid, chicken litter with straw bedding, chicken litter with sawdust bedding, pig litter with straw bedding or municipal waste compost. Letters indicate significant differences (p > 0.05). Error bars indicate standard error. n = 4. Adapted from Mackay et al. (2017).
Experiment Two
Increasing amounts of phosphoric acid addition to soil resulted in greater plant P uptake until 15kg P/ha (Figure 2), which we concluded to be the optimal rate of P addition to this soil. When phosphoric acid was applied at a below optimal rate (either 5 or 10kg P/ha) in combination with chicken litter (either at 5, 10 or 15kg P/ha), similar plant P uptakes were achieved as when phosphoric acid was applied at the optimal rate. Therefore, we conclude that combined use of OA and mineral P fertilisers can effectively meet crop P demands. However, further work is needed to validate these results on various soil types, with various mineral P fertilisers, and under field conditions.
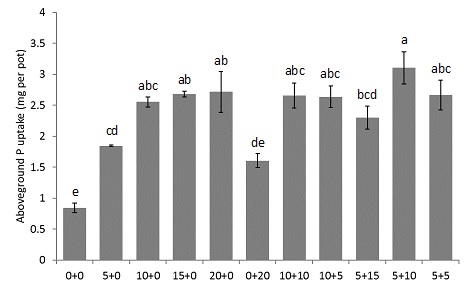
Figure 2. Above ground plant P uptake (content) per pot of plants grown in a greenhouse experiment where P was applied as phosphoric acid and/or chicken litter. The first number in the treatment name refers to the amount of P applied, in kg per ha, as phosphoric acid. The second number in the treatment name refers to the amount of P applied, in kg per ha, as chicken litter. Letters indicate significant differences (p > 0.05). Error bars indicate standard error. n = 4 (or 3 for 10+10 and 5+5).
Increasing amounts of phosphoric acid addition to soil reduced AM colonisation, with the lowest colonisation rates observed when phosphoric acid was applied at a rate of 20kg P/ha. However, when P was applied as a combination of phosphoric acid and chicken litter, colonisation rates were more than double compared to when just phosphoric acid was applied at the optimal rate (15kg P/ha). This indicates that substituting some of the phosphoric acid with chicken litter can dramatically improve AM colonisation. While the function of AM was not investigated in this experiment, it is well known that AM have multiple benefits, including improving soil structure (Gianinazzi et al., 2010). By incorporating OA into P management strategies, AM levels can be maintained.
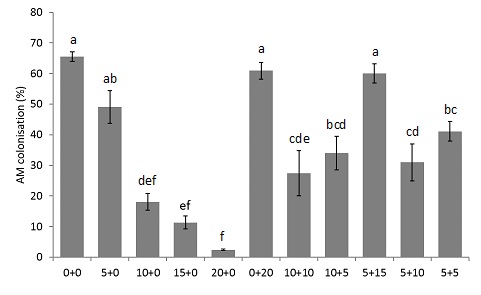
Figure 3. Percent of roots colonised by arbuscular mycorrhizas in plants grown in a greenhouse experiment where P was applied as phosphoric acid and/or chicken litter. The first number in the treatment name refers to the amount of P applied, in kg per ha, as phosphoric acid. The second number in the treatment name refers to the amount of P applied, in kg per ha, as chicken litter. Letters indicate significant differences (p > 0.05). Error bars indicate standard error. n = 4 (or 3 for 10+10 and 5+5).
Conclusion
The plant available P content of organic amendments can be adequately estimated by a bicarbonate-extractable method. This provides a better measure of P value of an amendment compared to total P. The animal litters and compost used in the study may not be suitable to meet crop P demands alone. Mycorrhizal colonisation declined with increasing mineral P fertiliser, but increased with increasing OA addition. Combined use of OA with fertiliser top-up aimed at meeting demand could prove a valuable approach with additional benefits to overall soil condition/health.
Useful resources
References
Abbott, L.K., Robson, A.D., De Boer, G., 1984. The effect of phosphorus on the formation of hyphae in soil by the vesicular-arbuscular mycorrhizal fungus, Glomus fasciculatum. New Phytologist 97, 437–446. doi:10.1111/j.1469-8137.1984.tb03609.x
Colwell, J., 1963. The estimation of the phosphorus fertilizer requirements of wheat in southern New South Wales by soil analysis. Animal Production Science 3, 190–197.
Gianinazzi, S., Gollotte, A., Binet, M.-N., van Tuinen, D., Redecker, D., Wipf, D., 2010. Agroecology: the key role of arbuscular mycorrhizas in ecosystem services. Mycorrhiza 20, 519–530.
Mackay, J.E., Macdonald, L.M., Smernik, R.J., Cavagnaro, T.R., 2017. Organic amendments as phosphorus fertilisers: Chemical analyses, biological processes and plant P uptake. Soil Biology and Biochemistry 107, 50–59. doi:10.1016/j.soilbio.2016.12.008
Pearson, J.N., Jakobsen, I., 1993. The relative contribution of hyphae and roots to phosphorus uptake by arbuscular mycorrhizal plants, measured by dual labelling with P-32 and P-33. New Phytol 124, 489–494.
Scholz, R.W., Wellmer, F.-W., 2013. Approaching a dynamic view on the availability of mineral resources: What we may learn from the case of phosphorus? Global Environmental Change 23, 11–27. doi:10.1016/j.gloenvcha.2012.10.013
Sharpley, A., Moyer, B., 2000. Phosphorus forms in manure and compost and their release during simulated rainfall. Journal of Environment Quality 29, 1462. doi:10.2134/jeq2000.00472425002900050012x
Acknowledgements
This research was funded by the GRDC via a Grains Industry Research Scholarship (grant number GRS10686) to Jessica Mackay and funding from the ARC to Timothy Cavagnaro (grant number FT120100463). Jessica Mackay also received support through an Australian Government Research Training Program Scholarship.
Contact details
Jessica Mackay
University of Adelaide, Waite Campus, Glen Osmond, SA, 5064
08 8313 6530
jessica.mackay@adelaide.edu.au
@jess_mackay13
GRDC Project Code: GRS10686,
Was this page helpful?
YOUR FEEDBACK
