Septoria tritici blotch rears its head in central and southern NSW (Northern)
Author: Andrew Milgate, NSW DPI, Wagga Wagga Agricultural Institute | Date: 28 Feb 2017
Take home messages
- The widespread occurrence of STB across southern NSW was exacerbated by the unusually wet winter of 2016.
- The risk to crops in 2017 is higher than average with inoculum carry over expected to be high but disease development will be dependent on rainfall and timing of sowing.
- The fungicide resistance status of the NSW population will be provided during the update
- Current popular varieties are vulnerable to infection and yield loss under favourable disease conditions.
- Fungicide resistance management strategies need to be implemented on farm.
- Integrated disease management complements fungicide resistance management.
- To slow fungicide resistance do not apply the same triazole active ingredient more than once in a season.
Background
Septoria tritici blotch (STB) is a disease of wheat caused by the pathogen Zymoseptoria tritici and is of global importance to wheat growers. This pathogen causes significant yield losses and is difficult to manage because it has developed resistance to fungicides in most major wheat production areas around the world. In Australia STB resistance to triazole fungicides was first detected in samples collected in Victoria and Tasmania during 2011-2012. Our ongoing monitoring of this fungicide resistance is detecting more changes in the STB population and we are researching ways to manage this evolving fungicide resistance problem.
The STB symptoms observed by growers in paddocks are the necrotic leaf lesions with obvious small black fruiting structures on the surface called pycnidia. By the time these symptoms are visible on the leaf the infection has been developing unseen for a long time, typically 14-28 days. The initial infections within a paddock come from spores produced on old diseased wheat stubble. This is how the pathogen survives between seasons, on the stubble from the previous year. After the infected stubble has undergone a period of weathering in the field the pathogen grows fruiting structures that release spores to infect the young wheat crops.
We can see from the pathogens lifecycle that it relies on spending several months sheltered within the wheat stubble. So as farming systems have progressed and adopted more and more stubble retention STB has benefitted. In addition to creating an ideal environment for the survival of the pathogen between seasons we also give the pathogen a ‘free go’ by choosing to sow a small number of wheat varieties across the farming landscape. This has the effect of allowing the pathogen to select the most adapted strains on those varieties eventually leading to resistant varieties giving the appearance of becoming susceptible overtime.
The large STB population that has built up in the high rainfall regions in recent years has required the use of fungicides to control disease levels and prevent yield loss. The use of these fungicides has had a direct impact on the pathogen by selecting strains that have lower sensitivity to triazole fungicides. Our research is focussed on identifying the different forms of the known genes involved in fungicide resistance present in the STB population and tracking their movement within and between regions. Furthermore we have conducted testing to reveal the level of resistance to fungicides, this will inform growers about future management choices to slow the decline in fungicide efficacy. In addition we also carryout integrated disease management trials to show the benefits of combining different control options.
Methodology
What we have done is conduct surveys across the winter cereal high rainfall regions of Australia and made collections of STB where it occurs. Two types of analysis have then taken place on the isolated strains of STB. A molecular genetic approach where the cyp51 gene is sequenced to look for changes in the DNA code. Changes in the sequence of this gene can lead to altered folding of the protein that is targeted by triazole fungicides which in turn leads to loss of efficacy. The identified changes have then been compared to those associated with fungicide resistance in other part of the world. In addition a wet chemistry approach has also been used to determine the level of resistance in Australian strains of STB. This is where the strains are exposed to a range of fungicides and concentrations then their growth is measured to determine the effective concentration where 50% of growth is inhibited (EC50).
Results and discussion
Fungicide resistance
Triazoles
We have identified 11 changes in the cyp51 gene that result in changes to the protein it encodes. These result in 10 altered isoforms (different types) of the protein which interact with the triazole fungicides. All of these changes to the gene sequence have previously been identified in other regions of the world. However a number of the isoforms have not been previously described. Since our recent collections started in 2011 we have not identified any of what is considered the original sensitive (wildtype) form of the gene present in the Australian population.
Our wet chemistry tests support the molecular evidence that these changes do have an impact on the resistance to triazoles. The testing has revealed that there are a number of combinations which have elevated levels of resistance that will be noticeable to growers. The results are also demonstrating that if an isoform has high resistance to one triazole it will be high for all other currently available triazoles. However this does not mean that all triazoles will be ineffective in the field, this will be further discussed the sections below.
Regional patterns have emerged in the incidence of isoforms and the level of triazole resistance they convey. Tasmania has the highest levels of resistance when compared to Victoria and South Australia. This is due to the detection of a number of isoforms which are not prevalent on the mainland at this time. Tasmania has the greatest diversity of isoforms and is showing signs of more intensive selection for ones with higher levels of resistance. The resistance status of the southern NSW collections of STB will be presented at the update. At the time of writing this paper testing was in progress.
Strobilurins
No changes have been identified to the resistance status strobilurins in Australia. This mode of action is still effective and can be part of a fungicide management strategy for STB. It is worth noting that Australia is one of the few places in the world where strobilurins are still effective and we should be careful in managing this class of fungicides for the future. The strobilurins are at high risk of selecting for resistance when used repeatedly within a growing season.
SDHIs
With SDHI (Succinate DeHydrogenase Inhibitors) products becoming available in Australia it is worth noting that STB has developed resistance to this mode of action in the UK. So again this mode of action requires proactive management in order to maintain its usefulness. This means not applying it more than once in a season.
Fungicide resistance and disease management
To achieve fungicide resistance management and disease management there are three important steps growers need to implement
1) Stubble removal.
- Stubble is the source of the infection each year. By removing stubble before sowing through whatever means there is a substantial reduction of pathogen population size.
- This reduces all isoforms irrespective of resistance and reduces the initial establishment of disease.
- But to be effective the removal must reduce infected stubble to very low levels, ideally below 100kg/ha infected stubble remaining within a paddock.
- Do not sow wheat on wheat.
- Under high disease pressure a variety rated moderately resistant – moderately susceptible (MRMS) can reduce the leaf area loss by as much as 60% compared to a susceptible – very susceptible variety (SVS).
- Host resistance reduces all isoforms irrespective of resistance and reduces the need for multiple canopy fungicide applications.
- But resistance ratings do change so crops must still be monitored in season for higher than expected reactions and check each year for updates to disease ratings.
- Do not use the same triazole active ingredient more than one in a season. Do not use a strobilurin or SDHI more than once in a season.
- Aim for early control of disease. STB spreads up the leaf layers of the canopy through rain splash and direct leaf contact. Reducing the disease in the lower canopy slows the upward movement of disease and ultimately the leaf area lost.
- Follow label instructions at all times.
The suite of fungicide products available on the market specifically registered for the control of STB in the canopy or as up front treatments are limited. At the time of writing this article the modes of action are confined to the triazoles (epoxiconazole, tebuconazole, flutriafol, fluquinconazole, propiconazole, cyproconazole and triadimefon) and the strobilurins (azoxystrobin) and some are only available in combinations. All of the triazole active ingredients will be effective to some extent against most of the isoforms we have identified. However our testing has shown that the following ones will appear less so in Tasmania they are; triadimefon and cyproconazole. While epoxiconazole, tebuconazole, propiconazole, flutriafol and fluquinconazole have all shown shifts in resistance they will still control disease. Timing of application in the disease epidemic is critical to getting the most out of these products.
Integrated disease management
The following graph illustrates the benefits of combining variety resistance and fungicides or reducing the amount of stubble inoculum. In this scenario two varieties are contrasted, Axe which is susceptible to STB and Sunvex which is moderately resistant to moderately susceptible grown at Wagga Wagga in 2017 under dryland conditions.
Figure 1 shows the disease development within the canopy over time in 6 treatments. For Axe there are obvious reductions in disease with regular application of fungicides in the protected treatment. However when fungicides are applied only at GS31 under high disease pressure little impact is visible and the disease level remains high, similarly for the upfront treatment of fluquinconazole. Whereas when a 2 spray program of GS31+GS39 is applied control improves. Note that the effect of reducing inoculum in a susceptible variety has little impact on disease progress. When we examine the effect of the same treatments against a variety Sunvex with higher resistance the impact is very different. Now the benefits of fungicide applications are less obvious and reduction of stubble inoculum has a similar impact to the application of fungicides. This example shows that growers should look to using multiple strategies to reduce disease in their farming systems because the application of fungicides alone does not always result in improved yield outcomes in all varieties and all years.
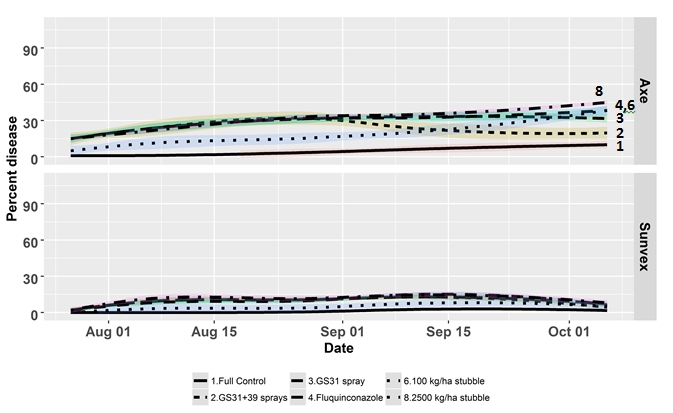
Figure 1. Trail grown at Wagga Wagga NSW 2017. Displayed here are a selection of varieties and treatments from the trial. The whole trial contained 5 varieties and eight treatments. The treatments displayed here are 1. Full control – multiple applications of propiconazole at 500ml/ha at 250 g/l active ingredient. 2. High disease pressure (2.5 t/ha stubble) with fungicide applied at GS31 and GS39. 3. High disease pressure with fungicide applied at GS31. 4. High disease pressure and fluquinconazole applied to the seed. 6. Low disease pressure (100 kg/ha stubble) 8. High disease pressure (2.5 t/ha stubble).
Conclusion
Understanding the lifecycle of STB presents opportunities for growers to be proactive about fungicide resistance and disease control. STB is a pathogen that survives from one crop to the next on the stubble left after harvest. During this period is an ideal time to take action and reduce the overall disease burden. In southern NSW during 2017 it is expected we will see infections of STB particularly in the higher rainfall areas and these will require action by growers to prevent losses. However in the lower rainfall areas early infections of crops may occur but without above average rainfall from August to October these infections will present lower threats to crops.
Submitting samples to fungicide resistance screening:The more samples of STB we receive the better informed growers are about the status of fungicide resistance.
To collect and submit samples please follow these instructions;
- Sample collection; Collect up to 20 leaves in total by walking in two parallel transects 10m apart collecting leaves which have STB lesions every 10m.
- Place the leaves in a paper bag and label with the following;
- Name, contact details, any fungicides applied to the crop and location
- Then post to
Dr. Andrew Milgate
WWAI, Pine Gully Rd, Wagga Wagga, NSW, 2650
Acknowledgements
Members of the cereal pathology team at WWAI who contribute to the fungicide resistance work; Melanie Renkin, Merrin Spackman and Beverly Orchard. Samples submitted to the survey by agronomists and growers across the HRZ and throughout central and southern NSW during 2016. The research undertaken as part of this project is made possible by the significant contributions of growers through both trial cooperation and the support of the GRDC, the author would like to thank them for their continued support.
Useful resources
GRDC Septoria tritici blotch Fact Sheet
http://www.extensionaus.com.au/minimising-fungicide-resistance-in-septoria-tritici/
Fungicide resistance talks at AARM 2015 – Session 1
Contact details
Dr. Andrew Milgate
WWAI, Pine Gully Rd, Wagga Wagga, NSW, 2650
Ph: 02 69381990
Email: Andrew.milgate@dpi.nsw.gov.au
GRDC Project Code: DAN00177,
Was this page helpful?
YOUR FEEDBACK
