Where did the low levels of Fusarium head blight come from in 2016 and what does it mean
Author: Steven Simpfendorfer(1), Daniele Giblot-Ducray(2), Diana Hartley(3) and Alan McKay(2) | Date: 28 Feb 2017
1NSW DPI, Tamworth; 2SARDI, Adelaide; 3CSIRO National Collections and Marine Infrastructure
Take home messages
- Low levels of Fusarium head blight (FHB) observed in central and northern NSW in 2016 were predominantly caused by Fusarium pseudograminearum (Fp).
- This was the crown rot fungus (Fp) reminding growers that it does not disappear in a wet season.
- FHB infection caused by Fp has reduced risk for mycotoxin accumulation in infected grain but could have detrimental impacts on crop establishment if retained for planting in 2017.
- Planting Fusarium infected grain can also introduce seed-borne crown rot infection into clean paddocks, undoing rotational benefits associated with growing non-host crops.
- Growers are urged to test both their crown rot inoculum levels in paddocks prior to sowing and ensure their 2017 planting seed has no or low levels of Fusarium infection if they observed FHB in 2016, especially if considering durum production.
Background
Above average rainfall was experienced in many parts of northern NSW in the 2016 winter cropping season. While this was great for increasing crop yield, unfortunately these conditions also favoured the development of a range of diseases. Of particular concern were low levels of head infections in durum and bread wheat crops and other concerns around black point and weather damaged grain. Fortunately these issues had a relatively low incidence in crops and were quite restricted in their distribution across the cropping region. However, during the actual flowering and heading stage of crop development misdiagnosis and associated unnecessary panic was not uncommon.
A survey of symptomatic heads and grain samples was conducted in 2016 to determine the various causes and to address concerns around Fusarium head blight (FHB) infections. FHB relates to the symptoms of head infection resulting in premature ripening of infected spikelets, generally caused by two fungi F. graminearum or F. pseudograminearum, following wet weather during flowering and/or grain-fill. White grain disorder, caused by Eutiarosporella spp. (formerly Botryosphaeria), produces similar visual symptoms that are not easily distinguished from FHB. These diseases are not uncommon in the northern grains region with the last widespread occurrence in northern NSW and southern Qld in 2010. NSW DPI conducted a similar study in 2010 with implications for mycotoxin production based on identification of causal species, issues with sowing infected grain and potential role of seed treatments presented at previous GRDC Updates. Some of this information will be covered in this paper as it is still very relevant to the situation that occurred in 2016.
Research in 2016
NSW DPI with the assistance of agronomists and growers conducted a survey of wheat crops with visible head infections or with discoloured white grains at harvest to determine the causal fungi. Head and grain symptoms were consistent with Fusarium head blight or white grain disorder so laboratory techniques were concentrated on recovery of these causal pathogens. Grain samples from seed sources targeted for sowing in 2017 were also assessed to determine the incidence of Fusarium and/or Eutiarosporella infection. Representative isolates collected from symptomatic heads or grain were identified to the species level using molecular techniques. Determining the exact causal pathogen has potential consequences for the risk of mycotoxin contamination and end use of affected grain.
What did we find and what does it mean?
Shot and sprung wheat
There were only limited reports of shot and sprung grain from the 2016 harvest in northern NSW. Damaged grain is usually downgraded to feed quality with no associated mycotoxin risks unless the grain goes mouldy which can occur if moisture content is above 12.5%. Feed value of shot and sprung grain can be reduced with bulk density being a good guide. Generally wheat with a bulk density <70 kg/hL, barley <60 kg/hL and triticale <67 kg/hL has around 60% the metabolised energy of good grain (Nourishing News, 2010).
Black point
Black point, which appears as a dark discolouration at the germ end of grain, is favoured by high humidity during the late stages of grain development. Hence, there were some instances in 2016 crops. Debate continues as to the actual cause of black point but definitive Australian research demonstrated that it is a physiological process related to the production of enzymes and not due to fungal infection. Either way, grain affected by black point is usually downgraded once above receival standards but is not associated with the production of mycotoxins as long as grain moisture content is maintained <12.5% to prevent the growth of moulds post-harvest. Black point is not desirable in durum wheat as it can result in undesired black specks in the pasta product but its impact on the quality of bread wheat is as debatable as its cause. Rees et al. (1984) in a study of the quality of black point affected grain stated “as the changes in quality detected were very slight, the condition had little effect on the value of the grain for bread making”.
Melanism
Initially FHB symptoms in wheat appear as small brown lesions on the glumes of infected spikelets within heads. Melanism (false- or pseudo-black chaff), is related to the overexpression of a brown pigment called melanoid under conditions of high humidity. The pigment concentrates in the glumes of wheat varieties (e.g. Suntop, Spitfire, Sunmate, Trojan) which carry the stem rust resistance gene Sr2 and was widespread in 2016. Unfortunately this physiological condition was sometimes misdiagnosed as early FHB infections. Melanism occurs on all glumes within a head and only discolours one side of the rachis (stem in head) so that when viewing from one side only every second segment is brown. Melanism can also cause the stem directly below the head to go brown and can even discolour stems in some varieties, with browning always extending downwards from a node. Initial FHB infections usually occur as point infections on one or two glumes within a head and when the infection progresses to the rachis it produces browning on both sides at that point. Fusarium infection can cause browning of the stem but crown rot symptoms generally extend up the stem from the tiller bases and when node infections do occur the browning always extends upwards not downwards from a node as with melanism.
Head infections
Head or grain samples were collected from a total of 80 paddocks from central and northern NSW in 2016 and causal pathogens identified to species. In 66% of cases FHB was caused by F. pseudograminearum (Fp) only, 4% by Fusarium graminearum (Fg) only, 19% were a mixed infection of Fp + Fg and 1% (one paddock) had a mixed infection from Fp and F. cerealis (Fcer)(Figure 1). A total of 4% paddocks had white grain disorder with recovery of Eutiarosporella (Eut) only with a further 4% having a mixed infection of Fp + Eut and 2% (2 paddocks) having mixed infection by Fp + Fg + Eut (Figure 1). Given the increased susceptibility of durum wheat to Fusarium infection, both FHB and crown rot, there was a slight dominance of samples coming from durum crops but plenty of infected bread wheat samples were also received.
Fusarium pseudograminearum (Fp) is the main species usually causing crown rot. Hence it appears that the low levels of FHB in 66% of paddocks surveyed in 2016 have come from Fp producing spore masses (macroconidia) on the lowest nodes of tillers infected with crown rot. Rain-splash then disperses these spores up the canopy to infect heads at flowering and cause low levels of FHB symptoms in a wet year.
There are two other main species of Fusarium which can cause FHB, being F. graminearum (Fg) and F. culmorum (Fc). Fg has more commonly been associated with FHB in the northern region and has a life stage (perithecia) which is produced on maize, sorghum, grass weeds and winter cereals. The perithecia are full of smaller spores called ascospores which are air-borne and hence more easily dispersed into wheat heads during flowering. Fortunately, Fp does not readily produce perithecia in the paddock and does not host on maize and sorghum. A total of 23% of paddocks had FHB infection associated with Fg which was most commonly in a mixed infection with Fp. Although Fc was not identified in any of the samples another species F. cerealis was identified in a durum sample from Terry Hie Hie in a mixed infection with Fp (Figure 1).
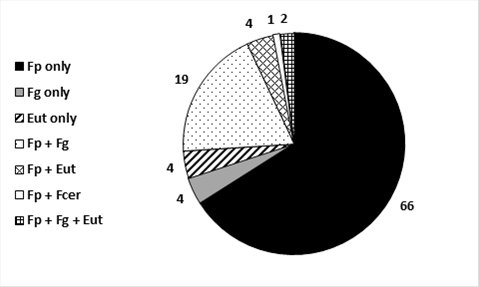
Figure 1. Fungal pathogens associated with head and grain infections in 2016
Fp = Fusarium pseudograminearum, Fg = F. graminearum, Eut = Eutiarosporella, Fcer = F. cerealis
Why is identifying the exact causal pathogen important?
In wetter seasons with frequent rainfall during flowering, which favour FHB infection, if it is Fp causing the infection then the incidence is usually considerably lower than if Fg is the causal pathogen. This is due to that lack of an air-borne spore stage (ascospore) with Fp, with prolonged wet weather required for Fp to first produce spore masses (macroconidia) around lower nodes on infected stems. The macroconidia, although still microscopic, are considerably heavier than ascospores hence they require splash dispersal to infect heads during flowering. This limits the dispersal of Fp. In contrast the ascospore stage in the life cycle of Fg is not as reliant on moisture for initial maturation of perithecia which contain the ascopsores. Rainfall during flowering is simply then required for the ascospores to be released, which then more readily are dispersed into heads during flowering by wind. This was consistent with the very low incidence of infected heads in paddocks in 2016 with most having well below 1% of heads visually infected.
Identification to species also has implications for potential mycotoxin issues within infected grain. The main mycotoxins produced by Fusarium are deoxynivalenol (DON) and nivalenol (NIV), with NIV being around 10 times more toxic than DON. DON is commonly called vomitoxin in the USA, with regulated limits of 1 ppm (1 mg/kg) in grain for human consumption, 5 ppm for pig feed and 10 ppm for beef/sheep/poultry feed. With Fg, DON levels are closely linked to the incidence of visually infected white and pink grains at harvest as mycotoxins are concentrated in these damaged seeds (Sinha and Savard, 1997). However, grain infected with Fp has been shown to accumulate much lower mycotoxin levels than that infected with Fg under laboratory conditions (Blaney and Dodman, 2002). This is supported by analysis of field samples from a previous occurrence of FHB in Australia in 1984 with Burgess et al. (1987) finding that grain with 38% Fp infection only accumulated 0.6 ppm of DON. There are also two different forms (chemotypes) of DON, with 3ADON being half as toxic as the 15ADON form. Similar research NSW DPI conducted following an FHB outbreak in northern NSW and southern Qld in 2010 determined that 92% of 137 Fp isolates examined were the 3ADON chemotype, 1.5% were 15ADON, 6.5% were 15 + 3ADON and none were NIV producers. In contrast 93% of the 88 Fg isolates examined were 15ADON, 3.5% were 3ADON and 3.5% were the NIV chemotype. Hence, determining which species of Fusarium is causing FHB is important as Fg generally produces larger quantities of more toxic forms of mycotoxins (NIV and 15ADON). Conversely, Fp, the main cause of FHB in 2010 and again in 2016 in this region, produces considerably lower quantities of a less toxic form of DON (3ADON) only.
Eutiarosporella spp. also cause a head infection with symptoms appearing as premature bleaching of spikelets and production of white grains. These symptoms are hard to distinguish from FHB. However, it has been shown that there are no mycotoxins associated with this pathogen and that grain infected with Eutiarosporella caused no issues when fed to weaner pigs for four weeks (Kopinski and Blaney, 2010). Hence, distinguishing Eutiarosporella infection from FHB has important consequences for the potential end use of affected grain.
Are there issues of retaining infected seed for sowing in 2017?
The issue with grain infection by Fusarium is that if it is sown the next year it can cause seedling death which reduces emergence. Crown rot infection can also be introduced to the base of surviving plants with infected grain also being an inoculum source for the infection of seedlings arising from uninfected grain. Grain infection with Fusarium only occurs as a result of FHB, which is favoured by wet conditions during flowering. Crown rot alone cannot directly result in grain infection, as the fungus does not grow up the entire stem and into heads within a season.
Additional trial work at Tamworth in 2011 investigated the effect of grain infection with Fusarium on emergence, and causing crown rot infection in surviving plants (seed-borne crown rot infection). Four seed lots naturally infected with varying levels of Fusarium (19 to 73%) during an outbreak of FHB in 2010 were used in the study.
Grain infected with Fusarium had lower emergence (only 15 to 55%) as it caused severe infection of the seedlings and many died, which is commonly called seedling blight. However, the trial also showed that plants which survived past the seedling-blight stage had also been infected with high levels of crown rot (average 35%). Seed-borne crown rot affects yield in the current crop and introduces infected stubble back into the paddock. Sowing Fusarium infected seed, therefore, undoes any break-crop benefits that may have been obtained from growing non-host crops (such as chickpea, canola, faba bean, sorghum) in the previous season.
Some seed treatments were shown to improve emergence of Fusarium infected grain by 10 to 30%, but had limited effect on reducing levels of seed-borne crown rot in surviving plants. Ideally growers should plant wheat seed that is free of Fusarium infection by targeting crops which were not infected with FHB in 2016 as their seed sources for 2017 plantings. Grain infected with FHB is usually white and, if prolonged wet conditions occurred during grain-fill, infected grains will take on a pink appearance. However, it should be noted that if any white or pink grains are evident, then the levels of Fusarium infection can be significantly higher than what may be indicated by visual inspection. This is because FHB infections that occur later during grain-fill may not cause any visual discolouration of the seed.
Implications
The low levels of FHB which occurred in bread wheat and durum crops across central and northern NSW in 2016 was predominantly related to infection by Fp. These infections arose from spore masses produced around lower nodes of tillers infected with crown rot which were then rain-splashed into heads during flowering. Mild conditions during Spring prevents the expression of crown rot as whiteheads. Consequently, crown rot infections often go unnoticed in wetter years. The low levels of FHB evident in 2016 could be viewed as the crown rot fungus (Fp) reminding growers that it does not go away in a wet season. Fortunately, the generally low incidence of FHB infection only resulted in a few instances where issues with the quality of harvested grain occurred in 2016. Hence, the overall economic impact of FHB was relatively minor in 2016. However, if Spring conditions in 2016 had been more stressed with limited rainfall and warmer temperatures during grain filling, then significant and widespread losses to crown rot are likely to have occurred. Growers need to not be complacent about potential crown rot inoculum levels leading into 2017. Avoid sowing winter cereals into paddocks which had FHB in 2016 as they are likely to represent a high risk for crown rot infection in 2017. All durum wheat varieties have increased susceptibility to Fusarium infection, both FHB and crown rot, hence durum production should be targeted to low risk paddocks preferably based on stubble or PreDicta B testing.
Growers who noticed or suspect that they had FHB or white grain disorder in 2016 should get their planting seed tested to determine infection levels prior to sowing in 2017. This information can be used to guide appropriate seed treatment options and to source cleaner seed with lower infection levels if required. This should be the preferred option compared to sowing seed of unknown Fusarium levels, which if moderate will result in poor establishment and introduce significant crown rot levels into paddocks. This will compromise rotational benefits that may have been achieved by growing a non-host crop in 2016.
References
Blaney BJ, Dodman RL (2002). Aust. J. Agric. Res 53: 1317-1326.
Burgess LW, Klein TA, Bryden WL, Tobin NF (1987). Aust. Plant Path 16:72-78.
Kopinski and Blaney BJ (2010). J. Anim. Phy. & Anim Nut 94: 44-54
Rees RG, Martin DJ, Law DP (1984). Aust. J. Exp. Agric. Anim. Hus 24: 601-605
Sinha RC, Savard ME (1997). Can. J. Plant. Path 19: 8-12
https://www.ag.ndsu.edu/pubs/plantsci/pests/pp1302.pdf
Acknowledgments
This research was undertaken as part of project DAN00176, which was made possible by the significant contributions of growers through both cooperation in sample collection and their support of the GRDC, I thank them for their continued support. Assistance from agronomists in collecting samples is also greatly appreciated. Technical assistance provided by Karen Cassin and Rachel Bannister is gratefully acknowledged.
Contact details
Dr Steven Simpfendorfer
NSW DPI
Ph: 0439 581 672
Email: steven.simpfendorfer@dpi.nsw.gov.au
Varieties displaying this symbol beside them are protected under the Plant Breeders Rights Act 1994.
GRDC Project Code: DAN00176,
Was this page helpful?
YOUR FEEDBACK
