Evolution of herbicide resistance in Barley grass populations across cropping systems of SA
Evolution of herbicide resistance in Barley grass populations across cropping systems of SA
Author: Lovreet Shergill, Benjamin Fleet, Peter Boutsalis, Christopher Preston and Gurjeet Gill | Date: 25 Feb 2014
Lovreet S Shergill, Benjamin Fleet, Peter Boutsalis, Christopher Preston and Gurjeet Gill,
School of Agriculture, Food & Wine, The University of Adelaide
Take-home messages
- We have reported the first cases of group A and B herbicide resistance in Hordeum glaucum in South Australia
- There are limited post-emergent herbicide options for the control of barley grass in crops. With the evolution of resistance to group A and B herbicides, barley grass control will become very difficult.
- Resistance to Group A herbicides in barley grass is associated with mutations in the target-site.
Introduction
Barley grass (Hordeum spp.) is a problematic weed that has been reported to be increasing in abundance in cropping systems in South Australia. It has been observed that management practices used in cropping systems in SA have selected highly dormant H. glaucum populations, which defer establishment until after the crops have been planted. Earlier, non-dormant populations of barley grass could be easily controlled with the use of knockdown herbicides applied in late autumn. This change in weed biology may have contributed to increased reliance on post-emergent herbicides for barley grass control in broadleaf crops. Previous research in SA had reported resistance to Group A (ACCase inhibiting) herbicides in H. leporinum. Current research undertaken at The University of Adelaide has confirmed the development of herbicide resistance in H. glaucum populations to Group A and B (ALS inhibiting) herbicides. In this paper we will report results of studies conducted to investigate the evolution of herbicide resistance in H. glaucum.
Herbicide resistance to Group A and B herbicides
Screening and dose response experiments were conducted, which have identified populations with varying levels of resistance to several Group A herbicides. Dose response studies have confirmed populations to be resistant to Targa® (quizalofop), Verdict® (haloxyfop) and cross resistant to Select® (clethodim). The repeated exposure of these populations to group A herbicides has resulted in the evolution of high levels of resistance (Figure 1). Sequencing of carboxyl-transferase (CT) domain of the ACCase gene from resistant plants confirmed target-site resistance as the mechanism of herbicide resistance. Sequencing from resistant plants confirmed the presence of previously known mutations at position 1781 in 5 populations and 2078 in other 2 populations.
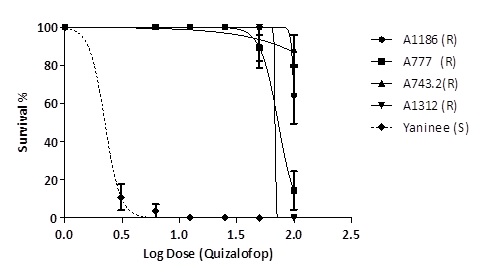
Figure 1. Effect of quizalofop on the survival of barley grass populations across SA and the susceptible population from Yaninee.
Following the confirmation of herbicide resistance in these populations, a random field survey was conducted to collect barley grass populations from the Upper North (UN) and Eyre Peninsula (EP) regions of SA in October 2012. Populations were randomly collected from cropping fields, pastures, scrub and fence lines. A total of 108 sites were sampled, but only 92 populations had enough seed to form a representative sample. In the following season, the populations were screened for herbicide resistance with Group A - Targa® and Group B - Raptor® (imazamox) and Intervix® (imazamox+imazapyr) herbicides at field rates. Survival assessments were taken at 28 days after application. Populations with greater than 20% plants surviving herbicide application were classified as resistant, whereas populations with 1 to 20% plant survival were classified as developing resistance. Where all plants were killed by the herbicide treatment, the population was classified as susceptible.
Table 1. Percentage of paddocks with Group A resistant barley grass in cropping regions of SA
|
Resistance Classification |
% Survivors |
Number of populations |
% of populations |
||||
|---|---|---|---|---|---|---|---|
|
UN |
EP |
Total |
UN |
EP |
Total |
||
|
Resistant |
> 20 |
5 |
2 |
7 |
24 |
3 |
8 |
|
Developing resistance |
1 – 20 |
5 |
2 |
7 |
24 |
3 |
8 |
|
Subtotal |
|
10 |
4 |
14 |
48 |
6 |
15 |
|
Susceptible |
0 |
11 |
67 |
78 |
52 |
94 |
85 |
|
Total |
|
21 |
71 |
92 |
|||
Across the surveyed area, 15% of the fields tested had a barley grass population with some level of resistance to Targa® (Table 1). As expected, there was a large variation in the percentage of resistant populations between the two regions. In UN region approximately half (48%) of the populations tested had some level of resistance, whereas, only 6% populations from EP exhibited some level of resistance to Targa®. Herbicide resistance was detected in samples collected from fields under crop (11 populations) and pasture (three populations), but cropped fields had a greater frequency of resistance.
Out of the 92 randomly collected populations tested with Raptor® and Intervix®, approximately 4% exhibited low level of resistance to group B herbicides (Table 2). All of the populations with Group B resistance were collected from the EP. Three of these Group B resistant populations were collected from wheat crops; in two paddocks there was a heavy infestation of barley grass but barley grass density was low in the third paddock. The fourth population came from a paddock sown with barley with thick patchy distribution of barley grass. Widespread adoption of ClearfieldTM technology has increased reliance on group B herbicides in local cropping systems, which may lead to greater occurrence of resistance to this herbicide group.
Table 2. Percentage of paddocks with Group B resistant barley grass in cropping regions of SA
|
Resistance Classification |
% Survivors |
Number of populations |
% of populations |
||||
|---|---|---|---|---|---|---|---|
|
UN |
EP |
Total |
UN |
EP |
Total |
||
|
Resistant |
> 20 |
0 |
0 |
0 |
0 |
0 |
0 |
|
Developing resistance |
1-20 |
0 |
4 |
4 |
0 |
6 |
4 |
|
Subtotal |
|
0 |
4 |
4 |
0 |
6 |
4 |
|
Susceptible |
0 |
21 |
67 |
88 |
100 |
94 |
96 |
|
Total |
|
21 |
71 |
92 |
|||
Acknowledgements
The authors are grateful for the financial support by ACIAR (John Allwright fellowship) for funding the PhD work. We also extend our gratitude to GRDC (UA00134) for partial funding of this research. Technical assistance by Rupinder Saini is also acknowledged.
Contact details
Lovreet Singh Shergill
The University of Adelaide, Waite campus, PMB 1, Glen Osmond, SA 5064
GRDC Project Code: UA00134,
Was this page helpful?
YOUR FEEDBACK
