Septoria fungicide control update and latest developments in rust management
Author: Nick Poole and Tracey Wylie (FAR Australia) | Date: 14 Feb 2017
Take home messages
- There should continue to be a focus on the principles of an integrated disease management (IDM) approach to control diseases such as STB, using rotation, cultivar resistance, later sowing, and other aspects of cultural control to complement fungicide control.
- New research on foliar fungicides indicates that the principal foliar fungicides still give good in-field control of STB up to 30 days after application when applied at full label rates.
- There are significant differences in disease control of STB and leaf rust amongst the different fungicide products and rates of application when monitored leaves are assessed more than 30 days after application.
- Single spray timings of foliar fungicide for control of STB made during the late tillering phase gave less effective disease control than applications made at first node (GS31). Spray application delayed until flag leaf gave the poorest control of STB but gave superior leaf rust control later in the season.
- Combining two adult plant resistance (APR) genes in Avocet Near Isogenic Lines (NILs) reduced the maximum yield response to stripe rust control from (significant responses) 0.98 and 0.4t/ha where the single genes were used along down to non-significant response of 0.22t/ha where the genes were combined.
Background
The identification of Septoria tritici blotch (STB) pathogen mutations in the laboratory that reduce triazole fungicide performance has increased concerns over the management of this disease with fungicides in the field. Whilst previous papers have covered the increased need for an Integrated Disease Management (IDM) approach to controlling this disease (a need that has not diminished), this paper looks at the field performance of foliar fungicides for the control of STB. A new GRDC funded project put forward by the Regional Cropping Solutions Network for the High Rainfall Zone commenced in 2016 to look at understanding and evaluating the effectiveness of fungicides to manage both STB and leaf rust in wheat. The stubble borne disease STB has been more prevalent over the 2016 season, having been observed in many areas of SA and spreading further north in NSW, as well as southern Victoria and Tasmania.Managing diseases in cereals is about more than fungicides, not least because of the recent discovery of reduced activity of triazole fungicides against the Septoria pathogen in eastern Australia. These pathogen mutations are conferring the first stage of fungicide resistance to this disease and in Tasmania, where fungicide usage is greater and mutations more severe, it appears to be impacting the field performance of fungicides. For this reason alone, growers need to pursue an IDM approach to control diseases such as STB.
In further work on cereal rusts, the paper reports on the influence of known APR genes and their influence on stripe rust necrosis when combined with a triazole/strobilurin fungicide mixture. This work has been conducted on NILs which allows the influence of the gene to be assessed in a common genetic background.
Research conducted in 2016
Field research was conducted at three sites in the 2016 season — Gnarwarre in southern Victoria, Hagley and Lake River in Tasmania and Conmurra in south east South Australia. All sites were subject to above average rainfall in the spring period, with rainfalls in excess of 100mm in September 2016. All three sites developed STB early in the season and leaf rust later in the season with the heaviest infections occurring at the Gnarwarre site in southern Victoria (the focus of this paper). Fungicide efficacy was evaluated in two trial series. Trial series 1 examined fungicide products and rates in order to determine the protectant and curative nature of these fungicides against STB and leaf rust. Eight different fungicide products and rates in trial series 1 were compared in two spray programs where each fungicide treatment was timed at first node GS31 and then repeated again at flag leaf emergence GS39. The same field trials were conducted at all three sites. Trial series 2 evaluated the influence of early season spray timings comparing a single application of triazole (epoxiconazole— Opus®125ɸ) and a formulated mixture of triazole and strobilurin (epoxiconazole/azoxystrobin) available as the branded product Radial®. This work was conducted at the Gnarwarre site in southern Victorian.
ɸLabel indicates that Opus®125 is registered for control of leaf rust in wheat but is not registered for control of STB.
At the time of writing this paper, trials were in the process of being harvested or were not yet ready for harvest, therefore the following paper is based on the disease data collected in spring 2016.
Results and discussion
Fungicide performance against Septoria tritici blotch (STB) — Zymoseptoria tritici
Influence of fungicide product
It should be emphasised that the fungicide products tested, with the exception of experimental coded material, are all approved for wheat and were tested for control of STB and leaf rust that occurred together in this experimentation. It should also be emphasised that not all products have an individual label recommendation for STB control.
STB was present in the southern Victorian trial from the early tillering stage onwards (June) with a good base of infection at the time of the first application at GS31 on 19 September. The same fungicide product and rate was then repeated at flag leaf emergence GS39 on 7 October following a number of rainfall events (probable infection periods) between the two sprays. Assessment conducted 15 and 31 days after the flag leaf application revealed good control of STB infection on flag-1 from products tested at their highest label rate, however at 52 days after flag application there were significant differences in fungicide persistence (Figure 1).
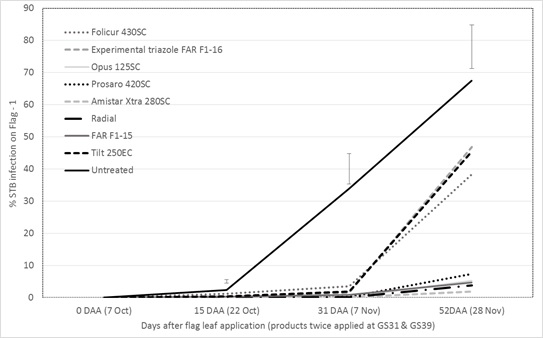
Figure 1. Influence of foliar fungicides approved for use in wheat on STB infection on flag-1 15, 31 and 52 days after the flag leaf application of a two spray program — cv SQP Revenue, Gnarwarre, Southern Victoria.
The difference in product performance apparent after 52 days on flag-1 was generally indicative of performance on lower leaves of the canopy assessed following full label rate fungicide application (Figure 2). There was evidence of significantly better curative control of STB on the lowest leaf (flag -3) from Opus 125® (epoxiconazole) and Prosaro® 420 SC (tebuconazole and prothioconazole), though it should be emphasised that though these products are approved for use in wheat, there is currently no label recommendation for STB control.
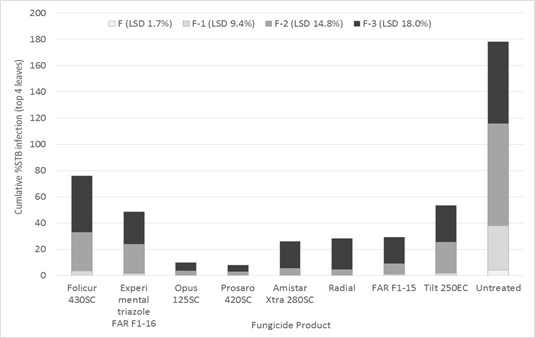
Figure 2. Influence of foliar fungicides approved (full label rate) for use in wheat on STB % infection on the top four leaves 31 days after the flag leaf application of a two spray program — cv SQP Revenue, Gnarwarre, Southern Victoria.
Influence of early season fungicide timing
STB is a stubble borne disease with the majority of the spore release from the stubble taking place in the autumn and winter under wet humid conditions. This initial spore release from the stubble is airborne and gives rise to the characteristic STB symptoms on the lower leaves of the crop later in the winter/spring. Further infection from these blotch lesions takes place under wet conditions with secondary spores spread up the plant by rain splash or the rubbing of wet leaves in the wind. These secondary spores are unable to travel long distances which means that the infection base you have in spring is likely to be your source of further infection, unlike the rusts where airborne spores can move into the crop later in spring. This raises the question as to when foliar fungicides should be sprayed in the spring to secure the best disease control and greatest economic response. To help answer that question, single applications of fungicide were applied at late tillering GS25 (17th August), pseudo stem erect GS30 (1 Sept), first node GS31 (16 September) and flag leaf emergence GS39 (13 October). Spraying early should control the disease at an early stage of the epidemic, although the leaves protected will be less important to grain fill. Spraying later allows greater early infection on the lower leaves, but applies fungicide to the first of the physiologically more important leaves for grain fill (flag-2 and flag-1). Figure 3 shows the influence of the different fungicide timings on disease through the course of the season.
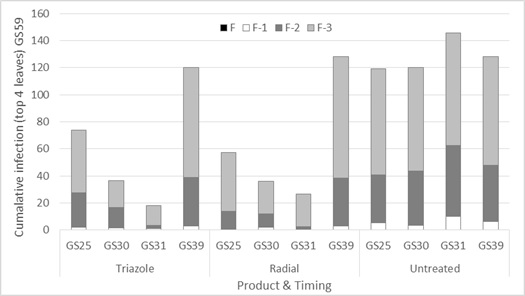
Figure 3. Influence of foliar fungicide mode of action and timing (triazole-epoxiconazole and Radial® that contains a mixture of two modes of action: triazole-epoxiconazole and strobilurin-azoxystrobin) on % STB infection on the top four leaves assessed at ear emergence (GS59 24 October) — cv SQP Revenue, Gnarwarre, Southern Victoria.
Results revealed that sprays applied at GS31 gave the optimum control of STB on the top four leaves when assessed at the ear emergence stage. This disease control was principally evident on flag-2 and flag-3. These two leaves emerged in the early September window.
Fungicide activity against rusts
Product performance against leaf rust — Puccinia triticina
2016 saw high levels of leaf rust build up later in the season in susceptible cereals in southern Victoria and Tasmania. The same work conducted on STB allowed the project team to evaluate fungicide performance against leaf rust which infected the crop later in the season compared to STB. Thirty one days after application, all products except the experimental triazole FAR F1-16 kept leaf rust infection below 5% irrespective of the rate tested (Figure 4). The stronger products against leaf rust in wheat were Amistar Xtra® (200, 400, 800ml/ha ɸa), Opus® (125, 250, 500ml/ha ɸb), Radial® (210, 420, 840ml/ha ɸc) Prosaro® (75, 150, 300ml/ha ɸd) and Folicur® at 145 and 290ml/ha. The efficacy was much reduced at 52 days after flag leaf application with 100% doses of Amistar Xtra®, Radial® and Opus® still delivering some level of control. The weakest products against leaf rust were Tilt® and the experimental triazole FAR F1-16.
ɸaLabel rate is 400 to 800ml/ha; ɸbLabel rate for leaf rust is 500ml/ha but for other diseases is 250 to 500ml/ha;
ɸcLabel rate is 420 to 840ml/ha; ɸdLabel rate is 150 to 300ml/ha.
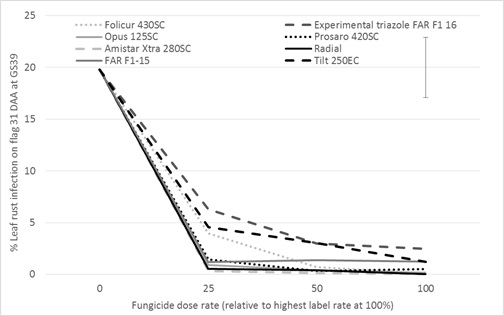
Figure 4. Influence of foliar fungicides approved for use in wheat at 25, 50 and 100% dose rates on % leaf rust infection on the flag leaf 31 days after flag leaf application — cv SQP Revenue, Gnarwarre, Southern Victoria.
Value of combining more than one APR gene against stripe rust in wheat
With newer foliar fungicides that confer greater green leaf retention one of the frequently asked questions is ‘do they have a value with cultivars protected by Adult Plant Resistance (APR)?’ FAR Australia in collaboration with The University of Sydney have been attempting to answer this question using both commercial cultivars and Near Isogenic Lines (NILs; genetically uniform lines that differ only in the presence of a rust resistance gene). Using NILs allows the influence of the APR gene to be assessed in a common genetic background. In work conducted at Wagga Wagga on the AGT trial site, NILs carrying different APR genes were treated with a combination of a triazole and strobilurin fungicide (Radial® epoxiconazole and azoxystrobin) at different development timings. The work funded under the Australian Cereal Rust Control Programme (ACRCP) revealed that the single APR genes Yr18, Yr29 and Yr46 significantly reduced a late infection (GS55-59) of stripe rust compared with the fully susceptible Avocet which is known to have no APR genes. The NILs with Yr29 and Yr46 single genes were more effective than Yr18 alone at preventing stripe rust necrosis (Figure 5). The combination of two APR genes in an Avocet background had significantly less stripe rust necrosis on the flag leaf than any of the single APR genes. The late onset of infection gave the APR genes the best opportunity to express their resistance to the disease given that APR genes are usually considered to be fully active by the flag leaf — ear emergence stage (GS39-59). With the late onset of infection the most effective spray timing was the single flag leaf emergence application, though this was only statistically superior to the head spray with NIL+Yr18 and susceptible Avocet. The early GS31 spray gave poor results because the infection onset was late and the leaves protected were flag-3 and flag-2 not the flag leaf and flag-1.
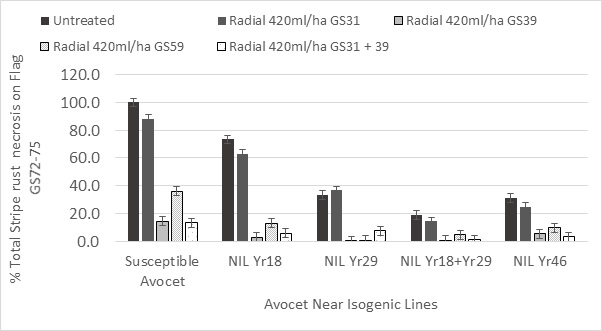
Figure 5. Influence of different APR genes Yr18, Yr29 & Yr46 in a common Avocet background on stripe rust necrosis during grain fill GS72-75.
The differences in stripe rust necrosis lead to the fungicide Radial® giving significant yield increases with all Near Isogenic lines except NIL Yr18+29 where two APR genes were incorporated into the genome (Figure 6). With NIL Yr18+Yr29 a single fungicide of Radial® at GS39 did significantly increase green leaf retention of the flag leaf at the doughy ripe stage (GS78-83) from 9% to 39% and increased yield by 0.22t/ha, however this increase was not statistically significant. With the exception of the fully susceptible Avocet there was no significant difference in fungicide response amongst the GS39, GS59 and two spray fungicide approaches. The early GS31 spray did not give a significant yield response over the untreated with any of the germplasm lines tested.
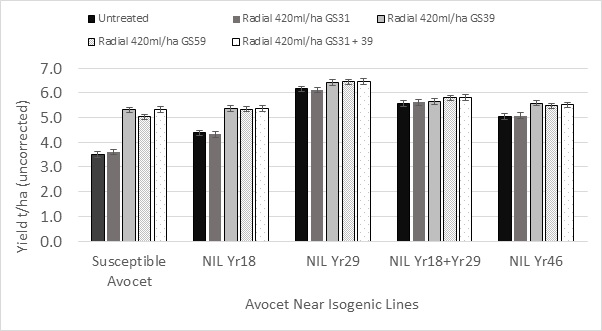
Figure 6. Influence of different APR genes Yr18, Yr29 & Yr46 in a common Avocet background on fungicide response from different application timings.
Conclusion
Foliar fungicides gave significant differences in STB and leaf rust control due to fungicide product, fungicide timing and fungicide rate, however the differences in disease control were most significant when assessed more than 30 days after application or at rates lower than the full-label rate recommended for wheat NILs with an Avocet background illustrated that two APR genes YR18 + YR29 combined, significantly reduced stripe rust necrosis compared to single APR gene effects of Yr18, Yr29 and Yr46. The differences in stripe rust necrosis lead to the fungicide Radial® giving significant yield increases with all Near Isogenic lines except NIL Yr18+29 where two APR genes were incorporated into the genome.
Acknowledgements
The research undertaken as part of this project is made possible by the significant contributions of growers through both trial cooperation and the support of the GRDC — the author would like to thank them for their continued support.
We would also like to acknowledge the work of our co-workers and collaborators in this project. For FAR 00004-a, Amanda Pearce and her colleagues in SARDI, Charlie Crozier at Mackillop Farm Management Group, Heather Cosgriff and her colleagues at Southern Farming Systems in Tasmania. For controlled environment work under the ACRCP program (FAR00002), Dr Karanjeet Sandhu at PBI Cobbitty and Dr Fran Lopez and his team at the Centre for Crop and Disease Management (CCDM) covering new actives research under Programme 9.
Contact details
Nick Poole
23 High St, Inverleigh, Victoria 3221
0499 888 066
nick.poole@far.org.nz
Was this page helpful?
YOUR FEEDBACK
