Successfully baiting round snails prior to egg laying on the Lower Eyre Peninsula in accordance to environmental conditions
Author: Jacob Giles and Blake Gontar (SARDI, Port Lincoln) and Michael Nash (formerly SARDI Entomology, Waite Campus) | Date: 01 Aug 2018
Take home messages
- Relative humidity >90% facilitates snail movement and feeding.
- Baiting is most effective when snails are actively feeding and when conducted before significant egg-laying.
- Summer weed control and cultural methods of snail control play a crucial role in managing populations where numbers are high.
Background
Research conducted on Lower Eyre Peninsula by Gontar and Nash in early 2016 found that by understanding the snail life cycle and reproductive activity, improved snail control could be possible. They found that hydrated and fed snails are capable of entering into a reproductive phase in March and may seek to lay eggs anytime from early April. This means that growers who wait to bait snails at seeding may miss the opportunity for control prior to egg laying.
In order to understand if active snails could be controlled through baiting during the January- March period (prior to traditional baiting practices) a trial was conducted at Mt Greenly on Lower Eyre Peninsula in 2017 and again in a similar nearby location at Frenchmans in 2018.
Methods
Baiting trials
In 2017 the field trial comprised replicated strips (2) of 1.5% metaldehyde snail bait. The bait was 2.5mm diameter and applied at 7.5kg/ha at two timings (6 February and 6 March) with untreated plots to account for natural population changes. Treatment strips were 50m wide, such that the resulting plots were 50m x 200m with 3 count points within each plot and 10 counts taken at each point. Initial population density for each plot over the trial was evaluated prior to baiting, with final counts taking place seven days post treatment.
Snail activity monitoring
A Brinno TLC100 time lapse camera was placed in the same field trial paddock, within one of the untreated control (UTC) replicates. The camera was located approximately 1m from the ground pointing vertically down. The field of view achieved was approximately 0.5 x 0.5m. The camera was set to take an image every 30 seconds. The camera was accompanied by an LED light bar connected to a 12 volt battery through a Kemo twilight switch, to allow the camera to take shots at night.
Immediately adjacent to the camera, a Hobo micro weather station was also established to closely monitor weather conditions at ground level. The weather station provided data on soil moisture, leaf area wetness and two sets of temperature and relative humidity data – one at ground level and the other approximately 40cm from the ground (i.e. at upper stubble height).
Analysis of the camera/weather data was undertaken by watching video and timing the snail activity each night. This was later matched with climate data.
Reproduction monitoring
From 2016 to 2018, 50 snails were collected regularly throughout the periods monitored. They were then preserved in ethanol until dissection at a later date. A subset of 25 snails were measured (shell diameter) then dissected, with the presence of food in their gut recorded, as well as the length of the albumen gland. The albumen gland of snails is known to enlarge relative to the size of the snail’s shell as the snail moves into a reproductive phase (Baker, 2012).
Results
Table 1. Snail population counts (snails/m2) before and after bait treatment on 6 February 2017. Population control represents the decrease in population that can be accounted for by baiting.
February 6th 2017 baiting treatment | Average | ||||||
|---|---|---|---|---|---|---|---|
Monitoring point | 1.1 | 1.2 | 1.3 | 2.1 | 2.2 | 2.3 | |
Snails/m2 before | 40 | 68 | 64 | 27 | 37 | 46 | 47 |
Snails/m2 after | 25 | 39 | 39 | 24 | 28 | 35 | 32 |
Population change | -0.38 | -0.43 | -0.39 | -0.11 | -0.24 | -0.24 | -0.30 |
Population control (%) | 0.27 | 0.31 | 0.29 | 0.08 | 0.18 | 0.18 | 22 |
Table 2. Snail population counts (snails/m2)before and after bait treatment on 6 March 2017. Population control represents the decrease in population that can be accounted for by baiting.
March 6th baiting treatment | Average | ||||||
|---|---|---|---|---|---|---|---|
Monitoring point | 1.1 | 1.2 | 1.3 | 2.1 | 2.2 | 2.3 | |
Snails/m2 before | 25 | 37 | 33 | 29 | 22 | 27 | 29 |
Snails/m2 after | 3 | 12 | 10 | 5 | 7 | 1 | 6 |
Population change | -0.88 | -0.68 | -0.70 | -0.83 | -0.68 | -0.96 | -0.79 |
Population control (%) | 0.62 | 0.48 | 0.49 | 0.59 | 0.48 | 0.68 | 56 |
Table 1 shows the poor response to baiting during the warmer period in February, which during 2017 was a period that did not support snail activity. During the week post baiting only two hours of significant snail movement was observed. Such low levels of snail activity are seen when conditions are dry, which is usually linked to hot, windy weather with a lack of rainfall. Baiting during such a period is ineffective.
Table 2 shows the snail’s mortality rate (measured as population control) as a result of baiting early March during ideal conditions. These conditions were linked mainly to dew events with only two hours due to rainfall at the end of this period and a total of 15 hours of substantial snail activity post baiting. This demonstrates the effectiveness of baiting when climatic conditions are suitable such as what was experienced during the week following the 6 March 2017. In March 2018 no significant control was seen due to a lack of snail activity throughout the baiting period.
Timing of baiting and risk
Snails commence egg laying once they have sufficient physical condition for breeding and there is sufficient soil moisture present to ensure their eggs will not dry out in the soil. To control the breeding population from reproducing, methods of control must take place prior to egg laying. Figure 1 illustrates a gradual increase in the size of a snail’s albumen gland from the beginning of autumn when environmental conditions begin to facilitate reproductive preparedness. From the commencement of this period until egg laying, snails will be active more consistently for greater periods of time in order to feed and gain condition. This activity correlates with cooler temperatures, more humid conditions and more frequent rainfall events.
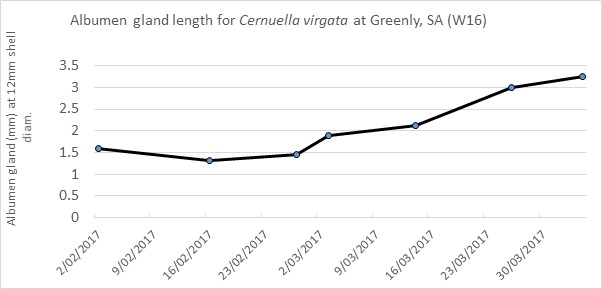
Figure 1. Average albumen gland size from 2 February until the 30 March 2017 at Greenly, SA.
During 2016 snails began to lay eggs on 21 April whereas in 2018 it was the 3 May. These dates when correlated with respective rainfall events demonstrate that snail egg-laying activity was triggered by surface soil moisture caused by rainfall events. It was observed that snails will take opportunities to lay come early April given their physical condition is suitable for reproduction. The exceptionally dry March in 2018 saw a laying date of the 3 May. This was 12 days later than in 2016 where around 60 hours of activity were monitored in the last two weeks of March allowing for sexual preparation and an earlier laying date.
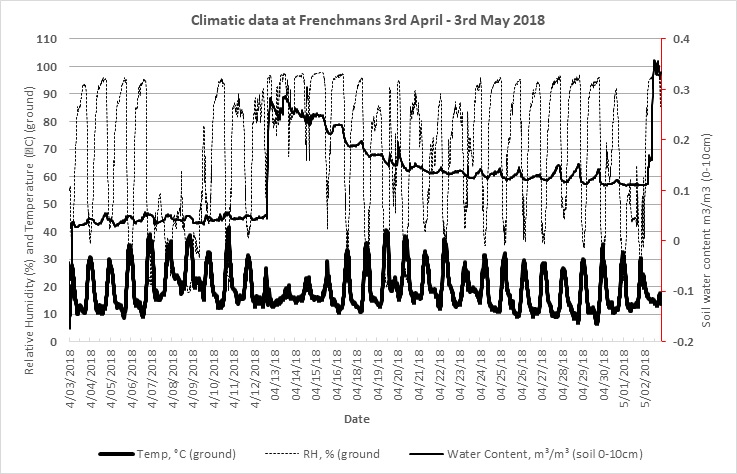
Figure 2. Ground temperature, relative humidity and soil moisture content (0-10cm) at Frenchmans, 2018.
Figure 2 displays climatic data obtained in a trial paddock leading up to egg laying on 3 May 2018. The high frequency of nights with a relative humidity >90% were conducive to high level of activity as snails prepared for egg laying. Growers paddocks observed on the Eyre Peninsula that were baited during mid-late April in 2018 saw high levels of control. Baiting on an ‘optimum’ forecast for snail activity on the 23 March 2018 saw no significant kill. This was due to a false weather forecast that actually resulted in heat and lack of rain, both of which are not conducive to snail activity.
Discussion
Cultural methods of snail control (i.e. cabling, rolling, etc.) have been shown to be highly effective at controlling snails (Brodie 2017). Such methods are well known to provide the best control on hot days when the chances of desiccation of snails is greater. If snails have food sources or the climatic conditions are not harsh enough (i.e. <35°C) to desiccate snails, then such methods are not as effective (Baker 2015).
Research conducted during this project shows that snails respond to climatic change rather than seasonal change throughout the year. This understanding of their behaviour provides growers with an opportunity to bait during periods of high activity prior to egg laying, which occurs following certain climatic conditions. The end result is more effective snail control via baiting. Egg laying is triggered by rainfall and once snails are sufficiently conditioned for breeding. High levels of activity are seen following a ground inversion (dew) or rainfall event that results in >90% relative humidity. A combination of surface soil moisture, low wind speed and day time temperatures below 30°C will greatly increase the chance of a ground inversion or dew. A heavy dew will facilitate around 8 hours of activity in a night. Bureau of Meteorology current weather observations, while not perfectly accurate are a very good guide to when relative humidity reaches >90%.
Baiting efficacy is further increased if ground cover and alternative food availability is decreased (Baker, 2015).
Conclusion
Cultural methods play an integral role in snail control over the summer months and should not be forgotten as they are a useful tool if implemented correctly. With increasing knowledge and a level of predictability in the weather, baiting is becoming another means by which snails can be controlled prior to egg laying in order to have an effect on a populations breeding potential going forward. However, if conditions do not facilitate snail activity, baiting efficacy will be compromised.
While snails must be active for baiting to be successful, if one waits too long for optimum conditions, snails will lay their eggs and the opportunity to control the breeding population prior to egg laying will be lost. Yearly differences between time of egg-laying, make determining when it’s best to bait more difficult and must be taken note of. If snails have been highly active through March they are more likely to lay earlier.
The level of risk involved in baiting prior to egg laying can be reduced by baiting closer to egg laying, however, not so close that the opportunity is missed.
Useful resources
BOM Current weather observations
References
Gontar, B and Nash, M. 2016, Improved Snail Management on Lower Eyre Peninsula, SARDI
Baker, G. 2015, Stubble retention: A barrier to snail management
Brodie, H. 2017, Integrated approach best for snail control, Nov-Dec 2017, GRDC Groundcover
Acknowledgement
This work was conducted on behalf of LEADA under the GRDC-funded Stubble Initiative (LEA00002).
The research undertaken as part of this project is made possible by the significant contributions of growers through both trial cooperation and the support of the GRDC — the authors would like to thank them for their continued support.
Thanks also to the Morgan and Parker families for providing and maintaining trial sites throughout the duration of the project and baiting where required.
Also many thanks to the SARDI team for assisting in data collection and analysis.
Contact details
Jacob Giles
jacob.giles@sa.gov.au
GRDC Project Code: LEA00002,
Was this page helpful?
YOUR FEEDBACK
