Best rotations for barley grass management
Best rotations for barley grass management
Key messages
- Herbicide resistance is evident in some populations but remains uncommon. There was no resistance to non-selective herbicides detected.
- The barley grass seed bank takes at least 3-4 years to control.
- Best control is achieved by break crops, with herbicides from different modes of action and control options to control late cohorts during the season.
- Crop competition can be effective for barley grass control.
Background
Barley grass is increasingly difficult to control, with a seed bank that lasts more than three years and which has staggered emergence. A national GRDC grower group driven project investigated optimal management strategies for local growers in Western Australia, South Australia and New South Wales (‘Demonstrating and validating the implementation of integrated weed management strategies to control barley grass in low rainfall zone farming systems’). The project ran from 2019 to 2021. In WA, the grower groups LIFT, SWEPA, KDG, MIG and FACEY/WANTFA investigated optimal pre-emergent herbicides, crop competitive ability, in-crop herbicides, and break crops for optimal barley grass control. Growers also identified fields where barley grass may have developed herbicide resistance. The management strategy highlights of the project’s third year are summarised below.
Method
Field trials
Each grower group identified a trial site on-farm to determine the best strategy for optimal control of barley grass over three years (Table 1). Each trial considered treatments ranging from low intensity (inexpensive) to high intensity (expensive) management options. Choice of crop rotation at each site was generally dictated by what each grower planned for the field in question. All trials were replicated, in a randomised block or split-plot design. Note: the WANTFA trial failed due to poor barley grass establishment in 2019 and was run in 2020 and 2021. Unlike the other sites, the WANTFA/FACEY trial was in a new site each year.
For each trial, the weed control treatments applied in 2021 (including herbicide and dates) are specified in the tables in the results section (i.e., Tables 2–5). Measurements included initial barley grass and crop density, barley grass head number, barley grass seed production and crop yield.
It is not the intent of this paper to present all three years of the project’s data from each of the five sites. Instead, the results and conclusions will cover highlights from the third year of each site. Full results of all trial sites can be found at GRDC Online Farm Trials (https://www.farmtrials.com.au/).
Herbicide resistance testing
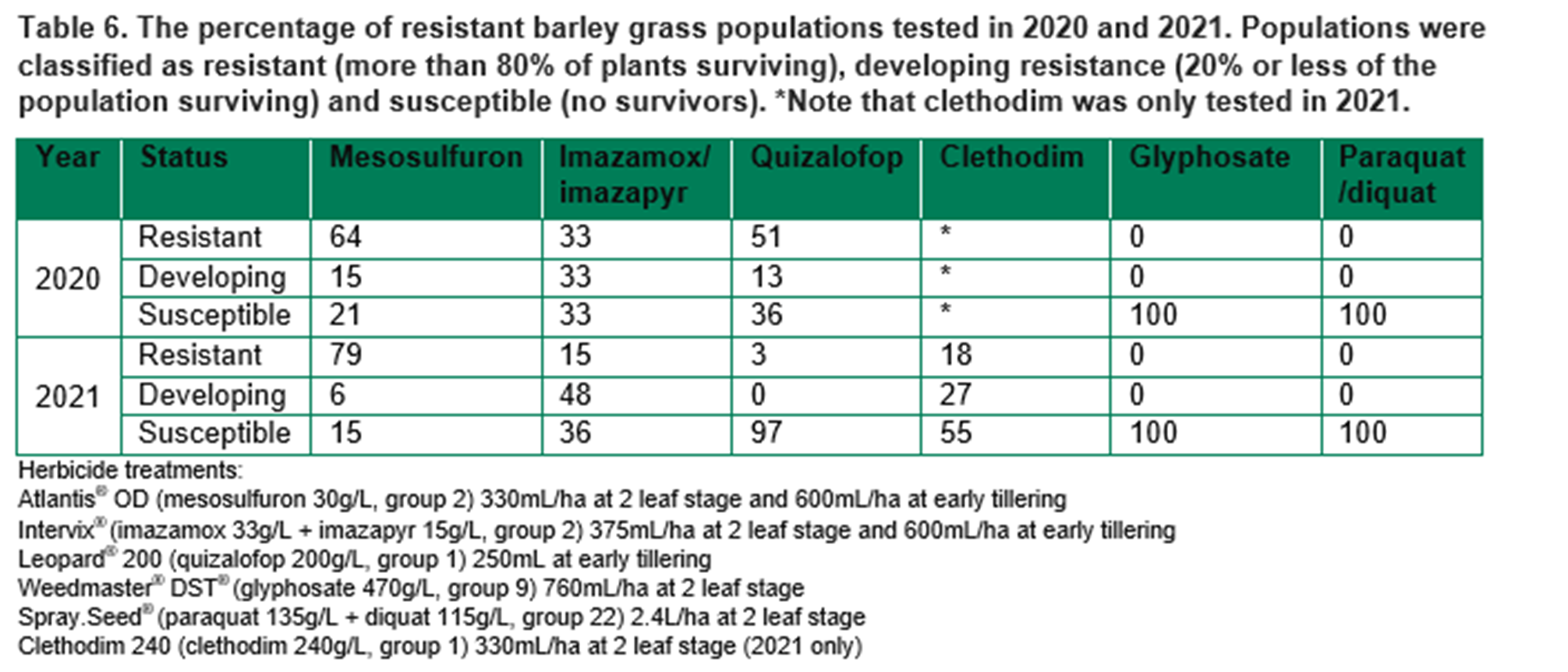
Resistance testing was conducted over 2020 and 2021. The testing was targeted rather than random, i.e., the samples were sent in because they were suspect resistant, rather than collecting random plant samples in the field. Barley grass seed samples were ‘after-ripened’ over summer and sown in trays of 15cm by 30cm by 5cm depth, with 20 seeds per tray. Herbicides (Table 6) were applied at the 2-leaf stage, early tillering or both. The herbicide testing methods were standardised across the project (i.e., uniform testing method for samples from WA, SA or NSW).
Results
Field trial - LIFT
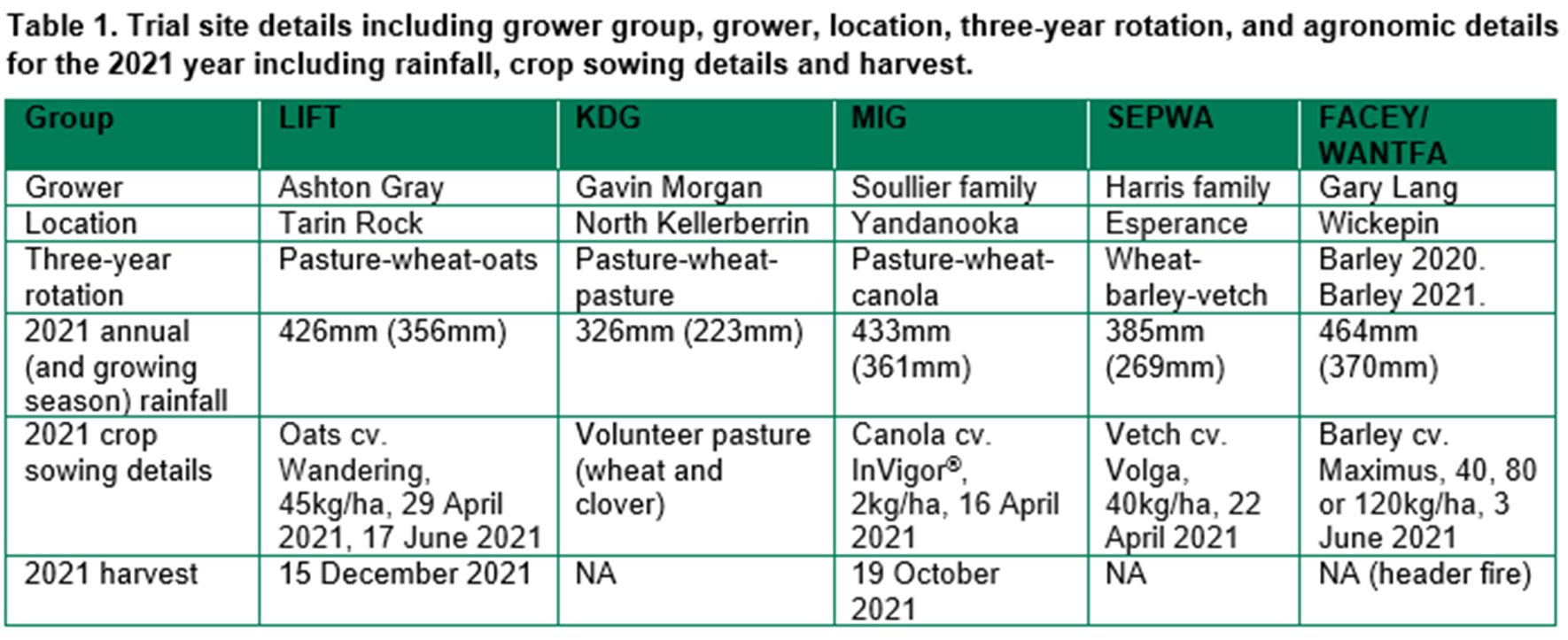
Barley grass control in the 2019 pasture included group 1 (A) herbicide or spray topping. Control in the 2020 and 2021 crops included early vs late sowing. Late sowing gave excellent weed control in 2021 (Table 2), with barley grass density close to zero. There were no panicles or seeds produced in the late sown plots. In the early sown plots, the number of barley grass plants, panicles and seeds were much higher, especially in the plots that were untreated controls during the pasture year of 2019.
Late sowing significantly reduced oat density (Table 2) as the very cold, wet conditions caused poor emergence at the later sowing date. As a result, yield was much lower in the late sown plots compared to those with early sowing. Within early sown plots, yield was reduced in the untreated plots where barley grass density was highest.
Field trial - KDG
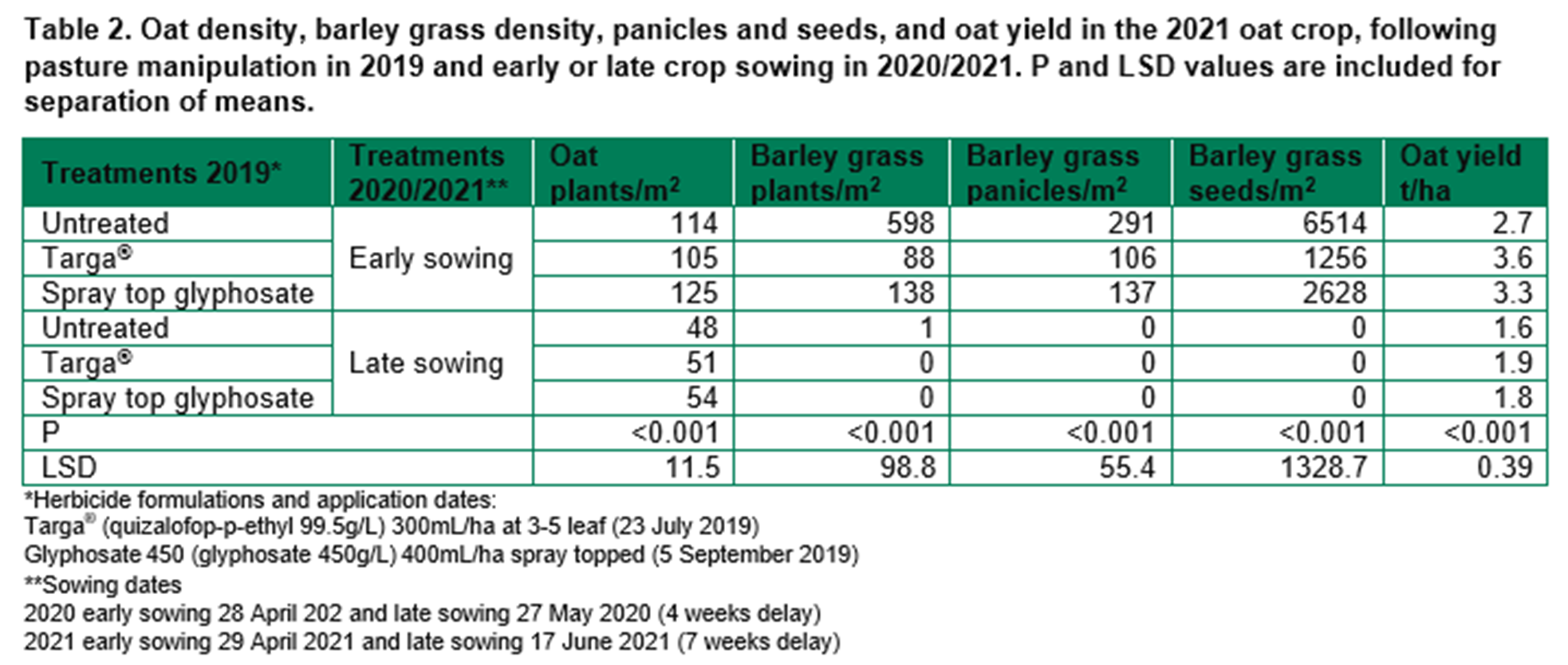
Barley grass control in the 2021 pasture included group 2 (B) herbicide or slashing. The reduction in barley grass density from group 2 (B) selective herbicide was not significant (Table 3). Slashing removed all panicles before maturity and prevented seed set. Pasture biomass was high in all treatments and was not affected by barley grass control.
Field trials - MIG
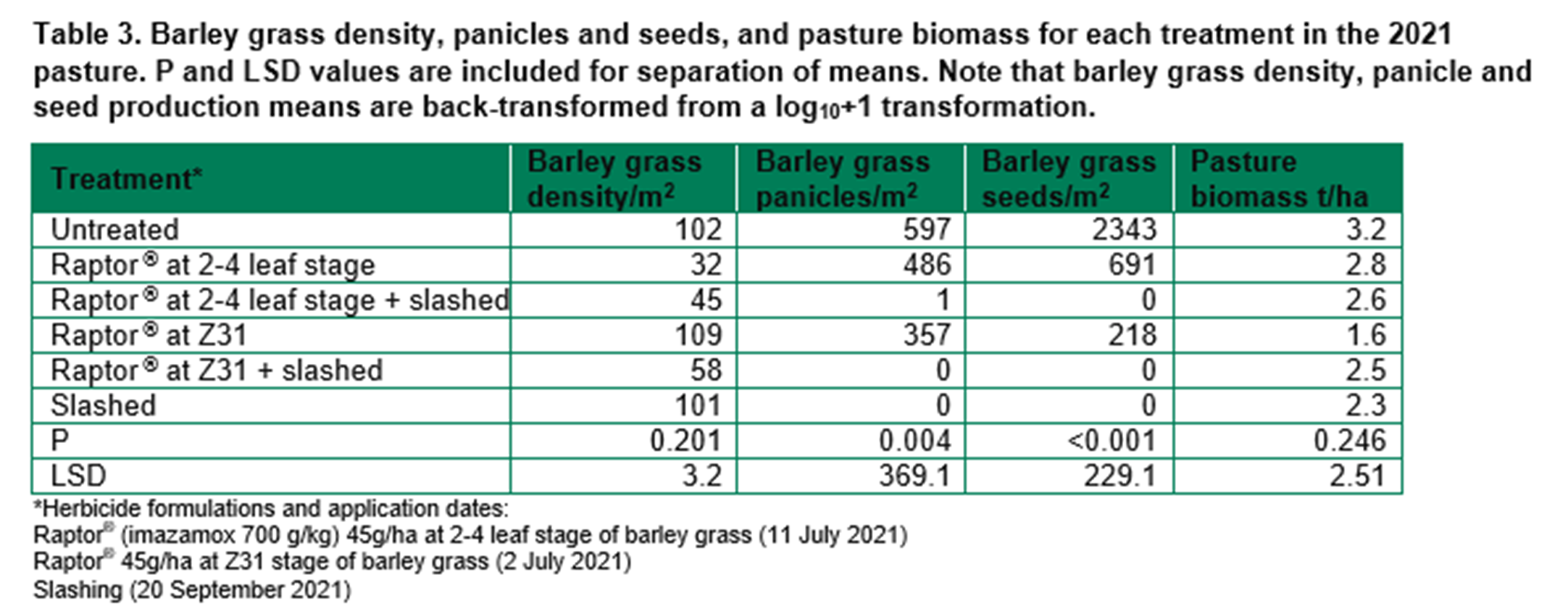
Barley grass control in the 2021 canola crop included pre- and post-emergent herbicides. Barley grass density was greatest in the treatment with Roundup Plantshield® alone (Table 4). Barley grass panicle number was too low to show a significant difference between treatments, but barley grass seed production was reduced in the treatments with TriflurX® or QPE compared to Roundup Plantshield® alone. The crop was healthy and highly competitive in 2021 due to high rainfall. The weed density was too low in all treatments to impact crop establishment or yield.
Field trials - SEPWA
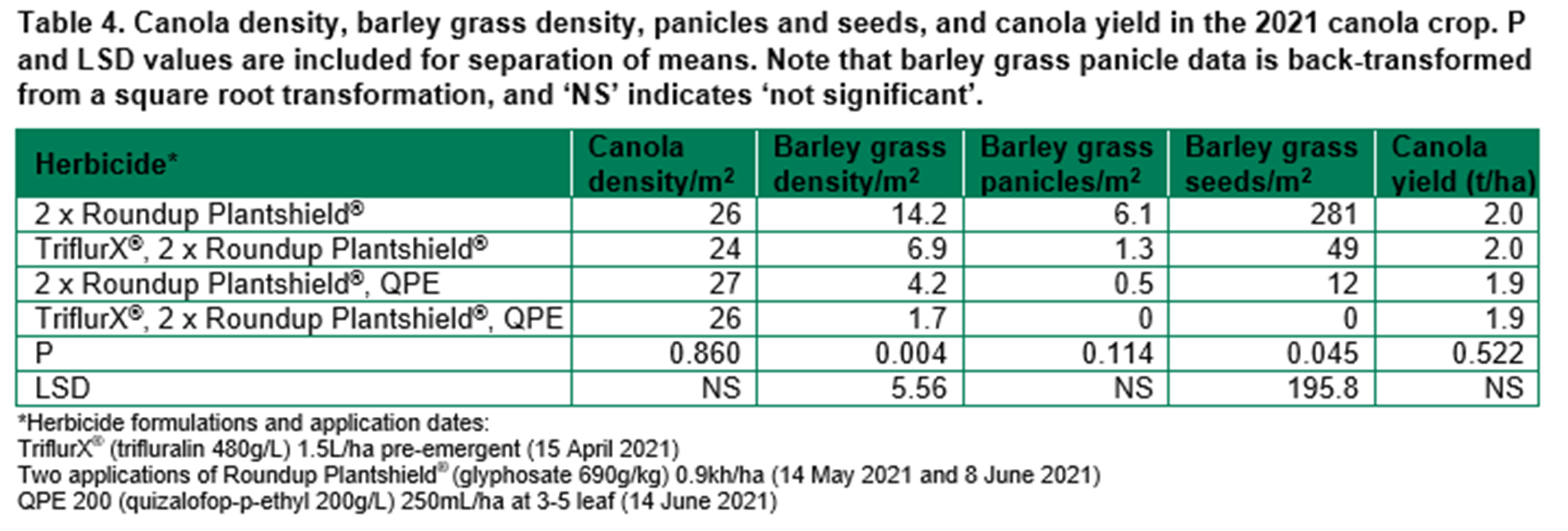
Barley grass control in the 2021 vetch pasture included pre- and post-emergent herbicides. There was a slight reduction in vetch density following Ultro® PRE, Quiz® + Clethodim® POST, but by the end of the year, there was no difference in pasture biomass (Table 5). Barley grass density in August was low in all treatments, but barley grass panicle and seed production were greatest following Treflan® PRE, Quiz® POST and close to zero in treatments with clethodim.
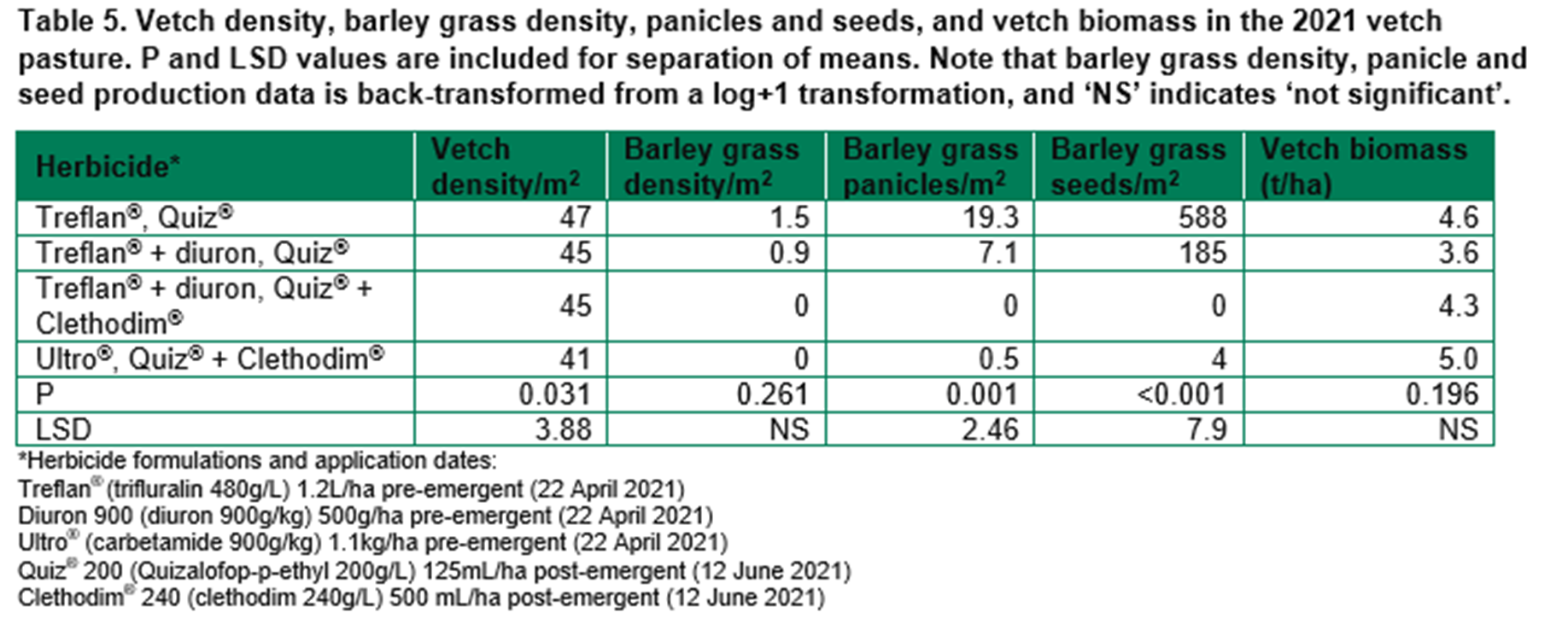
Field trials - WANTFA
Barley grass control in the 2021 barley crop included crop sowing rates of 40, 80 or 120kg/ha and Triflur X® (trifluralin 480g/L) at 2L/ha or 3L/ha. The original plan included Intervix® (imazamox 33g/L + imazapyr 15g/L). However, very wet conditions prevented in-crop herbicide application.
As expected, crop density increased with seeding rate (94, 159 and 200 plants/m2 at seeding rates of 40, 80 and 120kg/ha, P: 0.003, LSD: 34.4). Increasing crop density also reduced barley grass density (6.8, 2.3 and 2.5 barley grass/m2, P: 0.015, LSD: 2.01). However, further barley grass cohorts emerged late in the season. As a result, by the end of the season there was no difference in panicle and seed number. However, there was a consistent trend of reduced barley grass panicles or seeds at increased sowing rate (Figure 1).
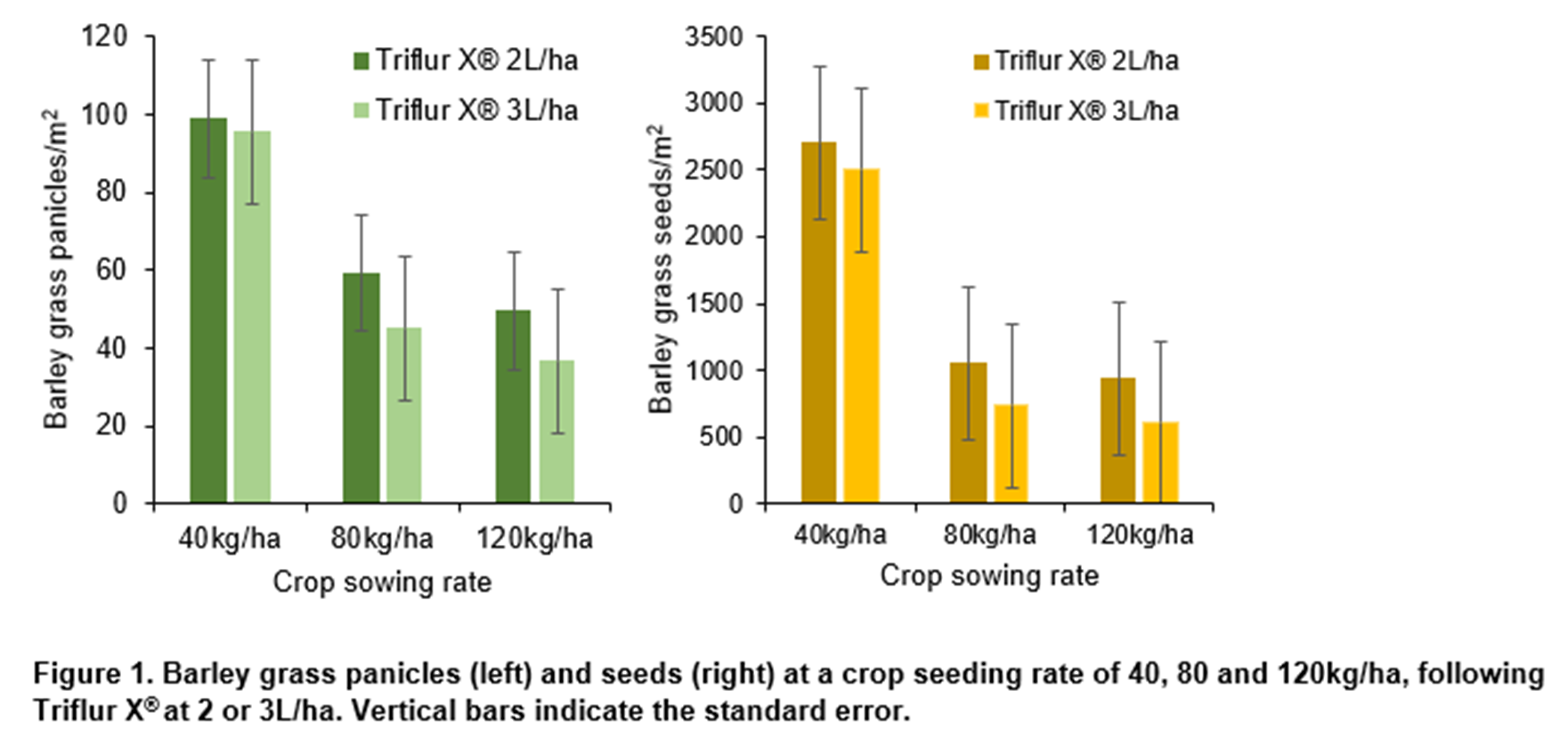
There was no resistance to glyphosate or paraquat/diquat in samples from 2020 or 2021 (Table 6). There was high resistance to group 1 (A) and group 2 (B) herbicides. However, all group 1 (A) and most group 2 (B) plants surviving those herbicides had stunted growth. Survival/seed production would have been reduced in field conditions.
Conclusion
There were no sites where barley grass was eradicated after three years of control. However, there were some common management messages from the five sites.
- The barley grass seed bank in 2021 was often high, after good control in 2019 and 2020. It takes at least 3-4 years (not two years) to reduce the seed bank.
- Break crops like pasture and canola allow for the use of a wide range of herbicides from different modes of action, and some late season control options to remove late barley grass cohorts.
- Delayed seeding is effective but can result in a large reduction to crop yield.
- Slashing is a highly effective control technique in pasture.
- Crop competition can be highly effective as a control measure, although it is influenced by barley grass height and competitive ability, which varies widely between ecotypes (Gill et al 2021).
Resistance to group 1 (A) and 2 (B) herbicides is developing, but there are still multiple fully susceptible populations. The current research was a targeted survey; the samples were sent in because resistance was suspected. Over the entire wheatbelt, resistance to group 1 and 2 herbicides remains uncommon in barley grass. However, resistance to group 1 and 2 herbicides as well as non-selective herbicides is increasingly common in SA and NSW, and these herbicides need to be used with care.
Acknowledgments
The research undertaken as part of this project is made possible by the significant contributions of growers through both trial cooperation and GRDC investment; the author would like to thank them for their continued support. We would also like to thank DPIRD project staff Nerys Wilkins, Dave Nicholson and Peter Gray and Professor Gurjeet Gill (UA), the national project leader. Thanks are due to Miranda Slaven for reviewing this paper.
Contact details
References
Gill, G., Borger, C. and Chauhan, B. (2021) Ecology of major emerging weeds. Kingston ACT, Australia: Grains Research and Development Corporation.
GRDC Project Code: UOA1903-004SAX,
