Minimising the impact of insecticides on beneficials in broadacre crops
Minimising the impact of insecticides on beneficials in broadacre crops
Author: Rosie Knapp (Cesar Australia), Luis Mata (Cesar Australia), Paul Umina (Cesar Australia & University of Melbourne), Melina Miles (DAF Qld), Ary Hoffmann (University of Melbourne), Lizzy Lowe (Cesar Australia) | Date: 22 Feb 2023
Take home messages
- Beneficials play an important role in supressing pest outbreaks in grain crops, but their populations are frequently diminished by the use of broad-spectrum insecticides
- ‘The beneficials chemical toxicity table for Australian grains' is now available to help growers and agronomists choose options that have a better IPM fit
- Increased biological control in crops can reduce the frequency of insecticide application required for pest control and minimise the risk of insecticide resistance evolving in grain pests.
Background
Biological control is an integral component of integrated pest management (IPM). Historically, the use of broad-spectrum insecticides in the grains industry has been widespread. In some crops, like canola, several insecticides can be applied between sowing and harvest. Predominantly, these have been synthetic pyrethroids, carbamates and organophosphates. These are indiscriminately highly toxic to all invertebrates and their use, particularly their repeated use, also depletes populations of beneficial insects such as the natural enemies of pests. This increases the risk of future pest outbreaks and hinders IPM programs.
Many insecticides have been registered over the last decade to control aphid, caterpillar and mite pests in the grains industry. Several newer products are promoted as selective towards pests and soft on non-target insects (and other invertebrates). However, little information is currently available on how ‘soft’ they really are on key beneficial groups. Cesar Australia and the GRDC have recently released a table that summarises the toxicity of foliar chemical sprays on a wide range of beneficials relevant to the grains industry. In this paper we discuss the research that went into developing this table and how this information and IPM practices can be applied on farms in south-eastern Australia in 2023.
Importance of beneficials in Australian grains
Globally, over-reliance on prophylactic chemical control has led to the emergence and spread of insecticide resistance in a range of crop pests. In the Australian grains industry, increasing resistance in key pests such as the diamondback moth (Plutella xylostella), redlegged earth mite (Halotydeus destructor), green peach aphid (Myzus persicae), and corn earworm/cotton bollworm (Helicoverpa armigera) exemplifies the need for a shift away from broad-spectrum chemical use. The preservation of beneficials such as insect and mite predators and parasitoid wasps to help control pest species is a central pillar of IPM farming approaches, aiming to reduce the industry’s current reliance on insecticides.
Beneficials make a valuable contribution in place of, or in combination with, chemical control in managing pest species. Beneficials in grains crops include generalist predators such as spiders, lacewings, ladybird beetles and damsel bugs, as well as parasitoid wasps such as Diaeretiella rapae, Lysiphlebus testaceipes and Aphidius colemani. When it comes to the control of aphid populations, farmers often use insecticides, but aphid parasitiod wasps and other insect predators have the capacity to suppress aphid populations and keep numbers below economic thresholds. Other examples of beneficials contributing to pest control include populations of redlegged earth mite and lucerne flea being consumed by predatory mites, including French Anystis mites and snout mites, and slugs being attacked by predatory beetles. Similarly, diamondback moths and other lepidopteran pests may be controlled by parasitiod wasp species such as Diadegma semiclausum, Apanteles ippeus, Diadromus collaris and Trichogramma spp., as well as by shield bugs, native earwigs, tachinid flies and spiders.
Impacts of current management practices on beneficials
The application of broad-spectrum foliar insecticides kills beneficials as well as pests. In some instances, this loss of beneficials from paddocks may lead to secondary pest outbreaks which would otherwise have been biologically controlled. This scenario is common and contributes to reliance on insecticides alone to control pest outbreaks. One of the easiest ways to reduce the impact of insecticides on beneficials is to use insecticides that have the least impact on these species (i.e., are the least toxic). Until now, the Australian grains industry has not had readily available information on the impact of insecticides on key beneficials. Between 2020 and 2022, Cesar Australia conducted standardised toxicity testing on many beneficials important to grains and assimilated this with existing studies into a toxicity table. This resource provides useful information growers and advisors need to make management decisions with preservation of beneficials in mind.
Methods
Generation of natural enemy toxicity data
Key beneficials and insecticides of importance to the Australian grains industry were identified through a literature review and industry consultation. Where knowledge gaps existed for particular beneficials or the results of previous studies were unclear, insecticide toxicity ratings through laboratory testing were generated following standardised protocols from the International Organisation for Biological Control (IOBC). In short, petri dishes were sprayed with insecticides at rates consistent with on-farm application at the maximum registered field rate (MRFR) in Australian grains. Once insecticide residue had dried (30-60 minutes), beneficials were added to dishes and their mortality observed over the next 2-3 days. This was repeated for 30 individuals of each species and survival percentages were calculated.
Development of the toxicity table
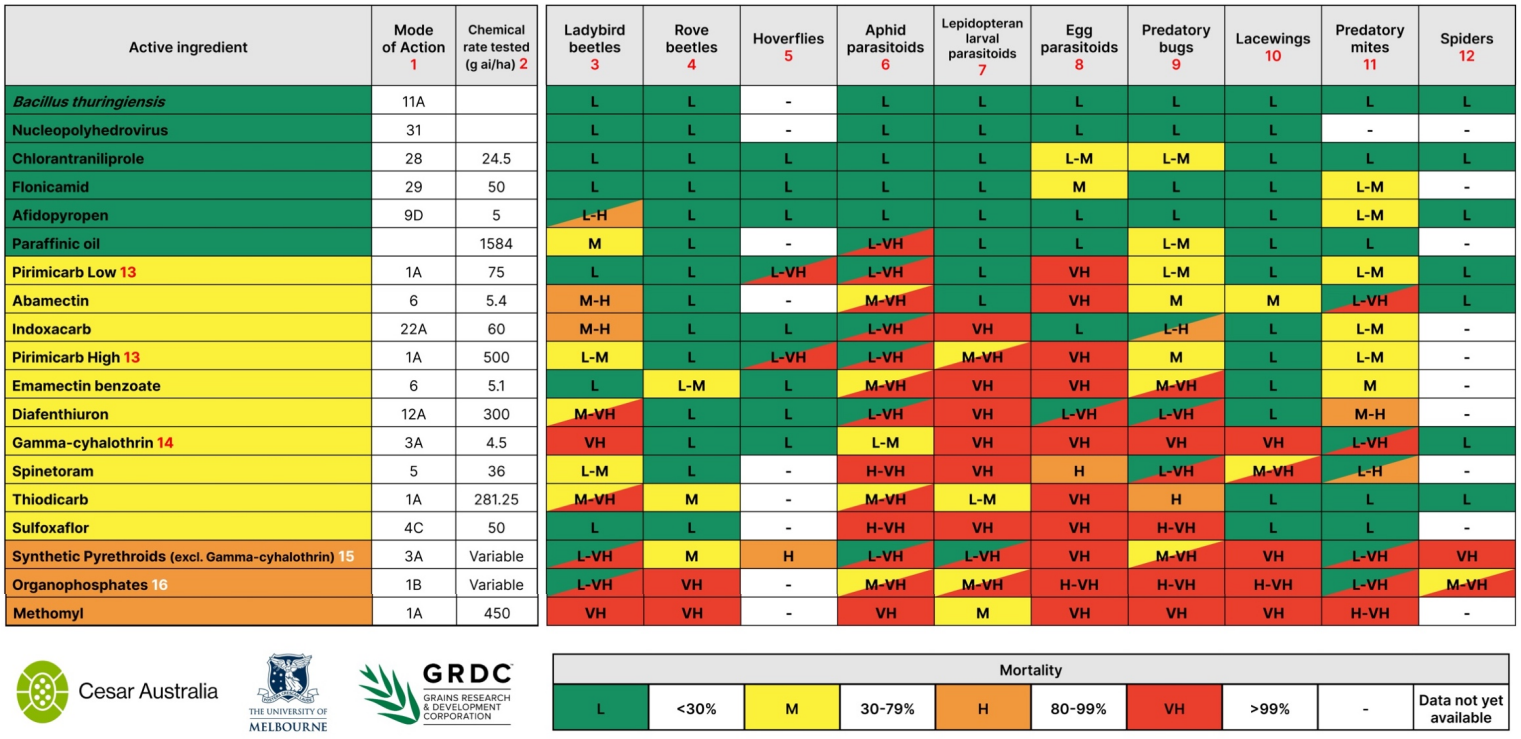
The resulting data was analysed, standardised and collated with previous research from Australia and overseas into a toxicity table. When needed, the data was condensed (e.g., by grouping related species) to make it a practical guide for growers and advisors. In the table, a rating of low (L) represents <30% mortality, medium (M) 30–79%, high (H) 80–99% and very high (VH) >99% mortality. These values represent mortality under controlled laboratory conditions – impacts in the field are likely to vary, especially if the crop gets multiple applications of insecticides. Where a range in toxicity was found among species or chemicals within a group, ratings are shown as cells with a diagonal slash, and the highest and lowest rating colours. The only natural enemy groups for which substantial gaps remain are those that cannot be obtained from commercial suppliers – namely, spiders and hoverflies. Research into these groups is ongoing, and subsequent revised table versions will be made available as these gaps are filled.
Results
In general, we found that active ingredients promoted as selective (or ‘soft’) produced low mortality rates (defined by the IOBC as <30% mortality) in beneficial species tested. This included the caterpillar selective chemical chlorantraniliprole, the newly registered aphid selective chemicals flonicamid and afidopyropen, and the bioinsecticides Bacillus thuringiensis and nucleopolyhedrovirus (NPV). These active ingredients are safe choices to use by growers dealing with caterpillars or aphids.
However, not all active ingredients marketed as soft on beneficials performed as expected. For example, despite being frequently promoted and discussed in the literature as a selective insecticide, pirimicarb was toxic to a range of beneficial species. Most striking is its very high toxicity (defined by the IOBC as >99% mortality) to some parasitoid wasp species, even at rates well below the MRFR. These parasitoids are key beneficials for the grains industry and make valuable contributions to the control of aphid pests. Pirimicarb application is therefore not compatible with IPM strategies seeking to make use of these beneficials in biological control.
A few of the tested species appeared particularly tolerant to a wide range of active ingredients, including rove beetles, hoverflies and spiders. These results show that certain beneficials may persist in a crop if it is sprayed with a relatively soft insecticide. This is encouraging for the future of IPM programs seeking to combine elements of biological and chemical control – if growers and advisers make careful selections in their choice of active ingredients, insecticides can still be used against pests with fewer detrimental impacts on their predators.
Table 1. The beneficials chemical toxicity table for Australian grains, published April 2022.
Use of toxicity table for informing management decisions
The beneficials chemical toxicity table for Australian grains (Table 1) is designed to assist growers and advisors in selecting insecticides that control pests while minimising the impact to beneficials. Where growers monitor and identify natural enemy species, spraying decisions can be made that preserve the biocontrol provided by resident beneficials. For example, if green peach aphids are building up and the grower wants to protect and encourage aphid parasitoids, the colour-coded mortality rating on the table makes it easy to see which chemicals to avoid, and which are safer for the parasitoid (e.g., pirimicarb could be replaced with flonicamid or afidopyropen). In situations where monitoring for beneficials is challenging, and knowledge of these species is limited, growers may select the overall least toxic chemical from the list (highest on the table) that is effective against the target pest. Table 2 gives examples of how changes in management practice of common pests in canola could have positive impacts on natural enemy populations.
Table 2. Examples of how softer pest management practices can benefit beneficials.
| Historic management practices | Softer alternatives | ||
|---|---|---|---|---|
Pest issue | Control method | Impacts to beneficials | Control method | Impacts to beneficials |
Redlegged earth mite | Organo-phosphates and synthetic pyrethroids | Very high toxicity to numerous beneficial groups, including predatory mites offering biocontrol | Cultural: Chemical: Diafenthiuron | None Reduced off-target effects on rove beetles, hoverflies, predatory bugs and lacewings |
Green peach aphid | Pirimicarb | Toxic to almost all parasitoids, as well as hoverflies, predatory bugs and predatory mites | Biological: Chemical: Flonicamid or Afidopyropen | None Low to medium toxicity to most beneficials |
Native budworm | Synthetic pyrethroids | Very high toxicity to almost all beneficial groups | Emamectin benzoate | Softer on ladybirds, rove beetles, hoverflies and lacewings |
Corn earworm/ | Synthetic pyrethroids | Very high toxicity to almost all beneficial groups | NPV | Soft on all beneficials |
Future research
More research is needed to quantify the contribution of biological control to pest management in grain crops, as well as the sub-lethal impacts of insecticides on beneficials. In addition, field studies that validate the laboratory-derived toxicity ratings will build industry confidence in this information. While we have investigated the effects of foliar sprays, we have not considered the impacts of insecticide seed treatments on beneficial species, which, based on overseas research, are expected to be considerable to many non-target groups.
Acknowledgements
The ‘Beneficials chemical toxicity table for Australian grains’ was developed by Robert McDougall (Cesar Australia), Kathy Overton (Cesar Australia), Ary Hoffmann (University of Melbourne), Samantha Ward (Cesar Australia) and Paul Umina (Cesar Australia), with support from the University of Melbourne and GRDC. We thank Jacquie Murphy, Karyn Moore, Lisa Kirkland, Marielle Babineau and Evatt Chirgwin for their assistance. The research undertaken as part of this project is made possible by the support of the GRDC, the authors thank them for their continued support.
Useful resources
Cesar Australia Beneficials chemical toxicity table (2022)
Cesar Australia blog post (2022) – Why should we care about beneficials in grans?
Cesar Australia blog post (2021) – Measuring the benefit of beneficials
Beneficial insects – the back pocket guide (2021) GRDC
Contact details
Associate Professor Paul Umina
Phone: +61 3 9349 4723
Email: pumina@cesaraustralia.com
Date published: February 2023
GRDC Project Code: UOM1906-002RTX,