Delivering weed-competitive, wheat breeding lines to growers
Delivering weed-competitive, wheat breeding lines to growers
Take home messages
- Increasing herbicide resistance, together with no new modes of herbicide action, are contributing to a ‘perfect storm’ in grain growers’ ongoing battle against weeds.
- The aim is to develop weed-competitive wheats to integrate with new and existing integrated pest management (IPM) strategies, thereby adding another option to the grower’s weed control toolbox.
- GRDC’s investment in weed-competitive wheat breeding began in 2005 delivering the first of the weed-competitive wheat germplasm. These lines showed up to 80% suppression in growth of annual ryegrass. However, take-up by breeders was small owing to agronomic and quality issues, together with lack of weed-competitive selection methods.
- Since 2005, new weed-competitive donors have been developed using the unique genetics from more than 30 overseas wheat varieties and landraces. These donors have approximately double the leaf area of today’s commercial wheat varieties.
- Dialogue with Australian breeding companies has identified main season, genetic backgrounds (i.e. the varieties Mace, Suntop, EGA Gregory, Magenta, Scout and Trojan) in the initial weed-competitive donors.
- The first selections out of an initial 7400 backcross-derived lines will be delivered to breeders in April 2018 for preliminary yield and quality testing. Subsequent crossing is underway into longer-season backgrounds including Kittyhawk, Pascal, Cutlass, and Flanker
- Breeding methods have been developed to assist breeders in selecting weed-competitive breeding lines in early and advanced generations: 1) A weed-competitive plant type with vigorous early growth, moderate-to-high tillering, and large flag/penultimate flag leaves, and 2) Plot-based, high-throughput phenotyping method using cultivated oats and barley as ‘competitors’.
Background
The cost of weeds to Australian agriculture is estimated at an annual $4 billion through crop yield loss and seed contamination. This estimate continues to rise with increasing crop value, and as herbicide resistance becomes increasingly more widespread. Currently, 25 different weed species display at least some level of resistance to widely deployed herbicides. Wheat is particularly vulnerable with the commonplace use of glyphosate as a chemical control of annual ryegrass and other weeds in direct drilling farming systems. Other herbicides may be used to assist in ryegrass control, but ryegrass has the capacity to develop resistance to other herbicide groups resulting in multiple genetic resistances. Indeed, herbicide resistant ryegrass is common throughout Australia and it is poor control of ryegrass that makes wheat the weakest link in weed management in Australian cereal rotations.
Wild oats, annual ryegrass and wild radish together contribute to significant reductions in wheat yields while adding to the growing weed seed bank with each wheat crop sown and harvested. The result of poor weed control is a reduction in wheat crop yields (of up to 90%) and potentially in the yields of subsequent crops. Consequently, it is estimated that herbicide costs are $80 to $100/ha for wheat, further reducing returns to growers. There are currently no new herbicide technologies that will assist in managing weeds and herbicide resistance in wheat. Integrated weed management (IWM) has proven to be economically beneficial in managing herbicide resistance and in minimising the size of weed seed banks over time. IWM employs a range of tactics for the effective, long term management of weeds e.g. pre- and post-emergent herbicides, bailing, windrowing and sowing competitive crops in rotation. Herbicides remain the most cost-effective weed management option, but herbicide resistance and the threat of few new herbicide chemistries suggests greater reliance on IWM approaches.
The potential for development of weed-competitive wheats was demonstrated in a previous GRDC project (UA61). Wheat lines developed from this project were assessed in multiple experiments across southern Australia and by different researchers. Assessment of the lines demonstrated the potential for breeding lines selected to be competitive to reduce and sometimes completely suppress weed species such as annual ryegrass and wild oats (Gill and Newman pers. comm.). Commercial wheat breeders showed some interest in this germplasm, but did not continue with breeding to include the traits owing to (1) a lack of understanding as to the need for weed-competitive wheat varieties in the marketplace, (2) uncertainty in the agronomic and grain quality in the developed germplasm, and (3) a lack of a repeatable high-throughput method in selecting for increased weed competitiveness in a commercial breeding program.
To overcome these shortfalls, the GRDC has invested in a new weed-competitive wheat breeding project aiming to develop: (1) New sources of improved weed competitiveness for use in breeding; (2) A robust and repeatable weed competitiveness screening methodology for use in commercial breeding programs; (3) Understanding of those morphological and physiological factors contributing to improved competitiveness; and (4) Release of spring wheat germplasm containing traits with improved competitiveness for use as parents in breeding of new weed-competitive varieties. Together, the project aims to deliver disease resistant, milling quality weed-competitive breeding lines into commercial breeding programs. These lines will be developed and tested in collaboration with commercial breeding programs throughout Australia, and in experiments validating trait and germplasm value conducted using multiple sites and years.
Strategy
A key feature of this project is the availability of new weed-competitive wheat germplasm developed in close engagement with Australian wheat breeding programs.
Germplasm
The previous CSIRO-University of Adelaide weed-competitive wheat project used elite, cycle 3, high vigour lines from the CSIRO high vigour recurrent selection program to produce breeding lines with Annuello, Wyalkatchem and Yitpi as parents, the genetic backgrounds. The recurrent selection population represented an aggregation of 30 diverse wheat lines with greater early vigour assembled from CSIRO studies (Rebetzke and Richards 1999). The recurrent selection program has continued since the cycle 3 germplasm available in 2006 to now include later cycles — cycle 4 to cycle 7 (Figure 1). These later cycles contain lines with substantially greater shoot and root vigour than for cycle 3 lines used as parents in the earlier weed-competitive wheat project. The evidence for this improvement is contained in Figure 1 showing the near-doubling in mean leaf area from parental germplasm to cycle 6 in four separate sowings (Zhang et al. 2015). It is noteworthy that the most vigorous of the Australian wheats (cv. Westonia) produced approximately half the leaf area of the most vigorous cycle 6 selection (compared with 51.2 versus. 87.3cm2).
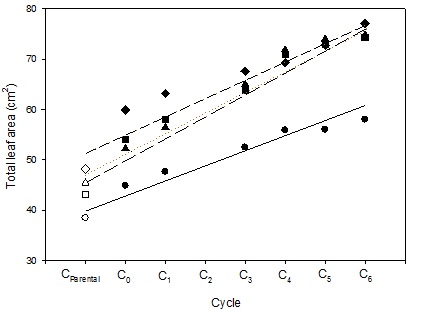
Figure 1. Relationship between cycle number and mean total leaf area measured in four environments: Sowing 1 (○; Y = 37.7 + 3.19 X, r2 = 0.93, P<0.01); Sowing 2 (□; Y = 41.8 + 5.16 X, r2 = 0.94, P<0.01); Sowing 3 (◊; Y = 48.5 + 4.24 X, r2 = 0.93, P<0.01); and Sowing 4 (∆; Y = 41.8 + 5.26 X, r2 = 0.95, P<0.01).
From the recurrent selection effort, four lines were identified from three separate cycles with significantly greater leaf width and leaf area: C4_32, C5_4, and C6_3 and C6_54. The basis for the greater leaf area was a wider leaf 1 and 2 allowing for a simple and quick surrogate for early leaf area and potential competitiveness.
A noticeable feature of lines developed from the high early vigour, recurrent selection lines is a larger flag and penultimate flag leaves (Figure 2). Greater early vigour and larger upper canopies have been reported overseas and in Australia as characteristics important in crop competitiveness (e.g. Huel and Hucl 1996; Coleman et al. 2001; Lemerle et al. 2001).
Figure 2. ‘Super-size’ flag leaf in weed-competitive CSIRO wheat breeding line W470201 (L) and commercial wheat Mace (R) at Roseworthy, SA, in 2016
Phenotyping methods for commercial breeding
The seed dormancy and establishment of ryegrass make creating uniform annual ryegrass pressure a real challenge in screening large numbers of breeding lines for weed competitiveness. The substitution of annual ryegrass with grain oats and barley as weed mimics that could easily be drilled with the wheat at sowing was investigated.
Figure 3. Comparison of barley growth (as a ‘weed mimic’) in Mace (L) and W470201 (R) at Yanco, SNSW in 2016.
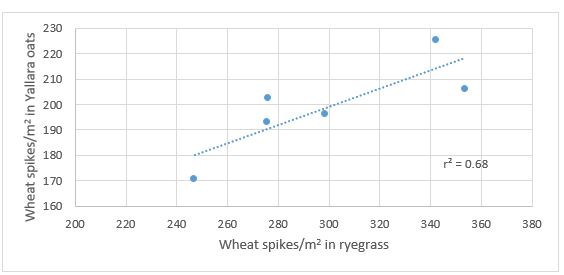
Figure 4. The positive relationship between mature wheat spike density in the presence of ryegrass and oats (cv. Yallara).
Commercial oats and barley establish well, are competitive and can be readily identified from wheat through the season (e.g. Figure 3) and then separated at harvest to establish measures of wheat suppression and tolerance of the competitor. The consistent ranking of different wheats in response to ryegrass and oats is clearly seen for an experiment at Roseworthy in 2016 (Figure 4). The ease in distinguishing wheat from barley competitors sown together is seen in Figure 4 where Mace struggles to compete with Fathom barley, whereas the larger flag leaf of the weed-competitive W470201 is seen to be suppressing barley ear number.
The value of a simple and reliable weed mimic is seen in Table 1, where the reduced competitive ability of commercial bread and durum wheats is contrasted with the suppression of more vigorous barley, triticale and CSIRO competitive wheat germplasm. Table 1 shows the potential of the CSIRO-developed germplasm to achieve higher yields in the presence of the oat competitor while maintaining reasonable yields in the absence of oats. Importantly, the relative grain yield in the presence of the oat competitor was typically high (>90%) for all vigorous cereals including the barleys and triticales (Table 1).
Table 1. Grain yield and growth scores for commercial cereal varieties and wheat germplasm grown with and without a commercial oats (as a weed mimic to simulate a competing grass weed).
Entry | Crop ground cover | Crop biomass at Z55 | Grain yield (with oats | Grain yield (w/out oats) | Relative yield | |||
|---|---|---|---|---|---|---|---|---|
(A) Bread wheat | ||||||||
| Axe | 55.9 | 722 | 3.14 | 3.81) | 82 | |||
| Cobra | 54.6 | 269 | 2.07 | 3.09 | 67 | |||
| Correll | 69.1 | 488 | 2.23 | 3.44 | 65 | |||
| EGA Gregory | 67.8 | 497 | 2.93 | 4.35 | 67 | |||
| Mace | 56.7 | 375 | 2.16 | 3.30 | 65 | |||
| Scout | 46.7 | 435 | 2.81 | 3.55 | 79 | |||
| Suntop | 64.4 | 632 | 2.29 | 3.53 | 65 | |||
| Spitfire | 65.7 | 708 | 4.23 | 4.90 | 86 | |||
(B) Durum wheat | ||||||||
| Caparoi | 63.8 | 532 | 1.97 | 2.97 | 66 | |||
| Jandaroi | 57.0 | 478 | 2.16 | 3.30 | 65 | |||
(C) Barley | ||||||||
| Fitzroy | 79.1 | 622 | 4.67 | 4.91 | 95 | |||
| Hindmarsh | 64.3 | 419 | 4.01 | 4.25 | 94 | |||
(D) Triticale | ||||||||
| Chopper | 59.1 | 597 | 4.08 | 4.11 | 99 | |||
Abacus | 72.7 | 471 | 3.34 | 3.63 | 92 | |||
(E) Selected weed-competitive wheat germplasm | ||||||||
CV117 | 66.5 | 498 | 3.36 | 4.01 | 84 | |||
WCD2-020213 | 62.8 | 520 | 3.79 | 3.95 | 96 | |||
WCD2-250310 | 70.3 | 614 | 3.61 | 3.82 | 95 | |||
WCD2-280504 | 80.1 | 724 | 3.41 | 3.62 | 94 | |||
WCD2-320101 | 69.5 | 697 | 2.74 | 3.75 | 73 | |||
WCD2-370303 | 67.2 | 1001 | 4.31 | 4.50 | 96 | |||
WCD2-390403 | 68.2 | 924 | 3.21 | 3.09 | 104 | |||
WCD2-470503 | 61.0 | 371 | 3.97 | 4.03 | 99 | |||
Av. LSD†† | 7.7 | 178 | 0.56 | 0.41 | 7 | |||
‡ Grain yield after removal of oat grain
† Percentage relative grain yield [i.e. 100*(grain yield with oats ÷ grain yield without oats)]
††Av. LSD is the least significant difference based on the average standard error of a difference
Deciding on wheat varieties and donors
The close engagement with breeders identified a set of commercial parents for crossing with the CSIRO developed germplasm as donors of the weed-competitive traits. The commercial parents initially included EGA Gregory, Espada, Magenta, Scout, Mace, and Suntop. A backcrossing methodology was employed to enrich populations for key agronomic and quality genes, and use of the new, alternative dwarfing genes Rht13 and Rht18 (Rebetzke et al. 2012). Introduction of Rht13 and Rht18 aims to maximise expression of cell and leaf size while maintaining plant and canopy height consistent with the commercial parents.
The final set of genetic backgrounds sown in 2017 included Cosmick, Crusader, EGA Gregory, Espada, Estoc, Mace, Magenta, and Scout and comprised approximately 7400 lines sown in Yanco, SNSW. Suntop and Trojan progeny were slower through population development and so approximately 3000 lines representing these parents will be sown in 2018. In 2019, a set of breeding lines in longer season backgrounds including Kittyhawk, Pascal, Cutlass, and Flanker will be sown for field evaluation.
A number of breeding companies (AGT, DAS, IGW, BCS) visited the Yanco nursery in 2017 to make decisions on what breeding lines were to be harvested for distribution back to the programs for agronomic and yield testing in 2018.
Exploring other methods of competitiveness
While the focus has been on increasing competitiveness through greater shoot growth, the project aims to explore other below-ground opportunities including a modified and more competitive root architecture in collaboration with Jairo Palta (CSIRO) and chemical control through allelopathy in collaborations with the weed ecology group led by Professor Leslie Weston (Charles Sturt University, Wagga Wagga, NSW).
Conclusion
The coupling of new genetics and improved phenotyping has developed weed-competitive wheat germplasm to be used as parents in the development of new varieties. The close engagement with commercial breeders throughout the extensive breeding effort is beginning to deliver large numbers of lines with their first yield assessments to commence in 2018.
When coupled with appropriate agronomy, harvest weed-seed collection technologies and new chemistries, the potential for competitive wheats to control weeds and reduce costs to growers is in the making.
Acknowledgements
The research undertaken as part of this project is made possible by the significant contributions of growers through the cooperation and the support of the GRDC. The authors would like to thank them for their continued support.
References
Coleman R.K., Gill G.S., Rebetzke G.J., 2001. Identification of quantitative trait loci for traits conferring weed competitiveness in wheat (Triticum aestivum L.). Australian Journal of Agricultural Research 52, 1235-1246.
Huel D.G., Hucl P., 1996. Genotypic variation for competitive ability in spring wheat. Plant Breeding 115, 325-329.
Lemerle, D., Gill, G.S., Murphy, C.E., Walker, S.R., Cousens, R.D., Mokhtari, S., Peltzer, S.J., Coleman, R. and Luckett, D.J., 2001. Genetic improvement and agronomy for enhanced wheat competitiveness with weeds. Australian Journal of Agricultural Research 52, 527-548.
Rebetzke G.J., Richards R.A., 1999. Genetic improvement of early vigour in wheat. Australian Journal of Agricultural Research 50, 291-301.
Rebetzke GJ, Ellis MH, Bonnett DG, D, Mickelson B, Condon AG, Richards RA 2012. Height reduction and agronomic performance for selected gibberellin-responsive dwarfing genes in bread wheat (Triticum aestivum L.). Field Crops Research 126, 87-96.
Zhang, L., Richards, R.A., Condon, A.G., Liu, D.C. and Rebetzke, G.J., 2014. Recurrent selection for wider seedling leaves increases early biomass and leaf area in wheat (Triticum aestivum L.). Journal of Experimental Botany 66, 1215-1226.
Contact details:
Greg Rebetzke
PO Box 1700 Acton Canberra Australia 2601
0429 994 226
Greg.Rebetzke@csiro.au
@rebetzke_0