Lime and liming – managing soil health
Lime and liming – managing soil health
Author: Lisa Miller (Southern Farming Systems) | Date: 26 Feb 2019
Take home messages
- Liming to maintain good soil pH levels and avoiding yield losses is just as important as applying fertiliser for maximising yields.
- If growers let soil pH levels in the topsoil run-down (pH in CaCl2 < 5.0) they are at risk of creating soil acidity issues at depth which are harder and more expensive to treat.
- In general, not enough lime has been applied frequently enough to address acidification occurring within the whole soil profile, so soil test to depth and calculate lime requirements for 0-10cm, 10-20cm and 20-30cm, not just the top 10cm of soil.
Background
Southern Farming Systems (SFS) was fortunate to be involved in a soil acidity project in both crops and pastures which started in 2014 under a GRDC and federal government investment in South West Victoria. Our understanding of soil acidity management from this work has been considerable regarding; lime responses, soil acidity increases in the 10-20cm layer, lime movement and lime quality. This work is set to be expanded in a new GRDC and NLP Smart Farms project across the high rainfall zone in Tasmania, Gippsland and in southern South Australia (SA). This paper reports on some of the SFS acidity research and related extension messages.
Importance of liming
Soil acidity affects major plant and soil functions
Soil acidification is unavoidable in productive farming systems, and acidity eventually eats away profits, affecting chemical, biological and physical functions within soils and plants. This makes it difficult to diagnose acidity based on crop symptoms. However, soil pH provides a good guide to which functions might be affected and the likely lime response as shown in Table 1.
Table 1. Crop symptoms at different soil pH (measured in CaCl2).
If the soil pH (CaCl2) is: | |
|---|---|
More than 5.5 | There will be no problems from soil acidity affecting crop growth and yield, and there could be net movement of lime beyond 10cm depth. |
Less than 5.2 | The effectiveness and numbers of rhizobia that fix nitrogen (N) on acid sensitive legumes (e.g. lucerne and pulses, but not narrow-leafed lupin) are reduced. Liming increases the persistence and effectiveness of these rhizobia, and the amount N fixed and grain produced of the sensitive legumes. |
Less than 5.0 | In addition to the effects above, there is a chance of molybdenum deficiency in legumes — check for local advice. Molybdenum is important in the synthesis of amino acids and proteins and a requirement for Rhizobium bacteria to fix atmospheric N. |
Less than 4.8 | In most soils, aluminium (Al) starts to precipitate from a harmless solid into a soluble form which is toxic to root growth. Aluminium tolerance among plant species varies. Reduced root growth means roots are unable to effectively explore soil for nutrients (particularly phosphorus and trace elements) and access stored subsoil water for growth or grain filling. Crop yield is reduced significantly. Reduction in root hairs occurs and so infection by rhizobia (nodulation of legumes) is severely affected. |
Less than 4.5 | The speed of N mineralisation processes (nitrification) slows significantly, resulting in decreased N supply. In most soils Al concentrations increase further and quickly become toxic to most pasture and crop species. There is a chance of molybdenum deficiency in cereals or canola, but check for local advice. The effectiveness of rhizobia in acid tolerant legumes, such as subclover, balansa and arrowleaf clover is reduced. |
Less than 3.8 | Soil can no longer buffer effectively against pH change and is overcome with acidity which breaks down clay minerals leaving only the sand component. Irreversible soil structural damage is done. |
(Source: Table adapted from Fenton, 2003)
Do not let soil pH run down
Our cars are serviced regularly so they run reliably and efficiently, and most people do not wait for their cars to break down and then have it fixed. The same applies to maintaining soils. If topsoil pH is allowed to run down to less than 5.0, which is common in grazing enterprises, significant production has probably been lost over the past 5-8 years and possibly not noticed. More importantly, by letting the soil acidity form in the top 10cm of soil, the acidity build-up results in leaching downwards of hydrogen ions and this allows soil acidity to increase at depths of 10cm to 20cm, where it becomes much more difficult and expensive to treat.
Lime is slowly soluble and often will not work straight away — it takes time to dissolve and move and so some ongoing yield losses will continue to be incurred. If a subsoil acidity problem exists, lime with no incorporation will take five years or more to fix the acidity profile beyond 10cm (depending upon soil type and rainfall), provided enough is applied to move downwards.
Maintaining good soil pH means yield responses to lime may not be immediately noticeable, but they will avoid ongoing acidification and yield declines. A soil pH increase will show that the lime is working, and regular soil monitoring is recommended, particularly at 10-20cm where there may be issues with subsurface acidity build-up.
Soil acidity eats away at yields
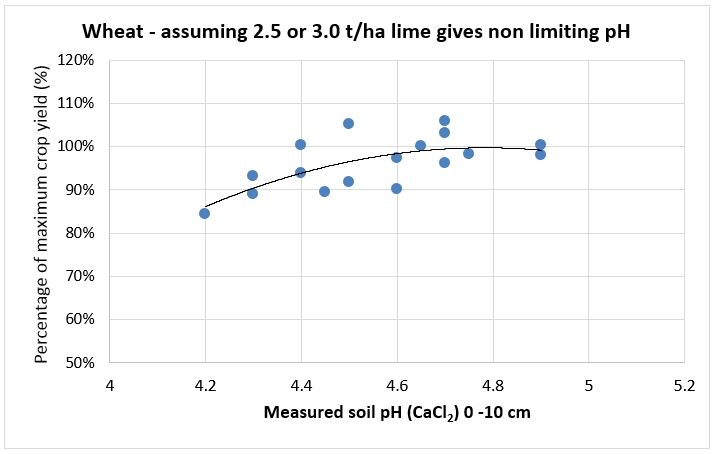
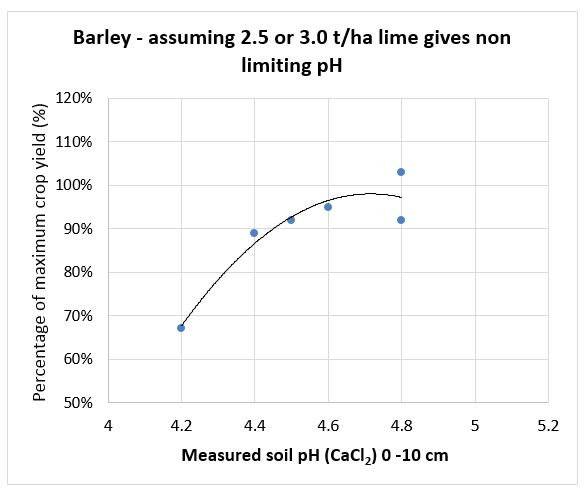
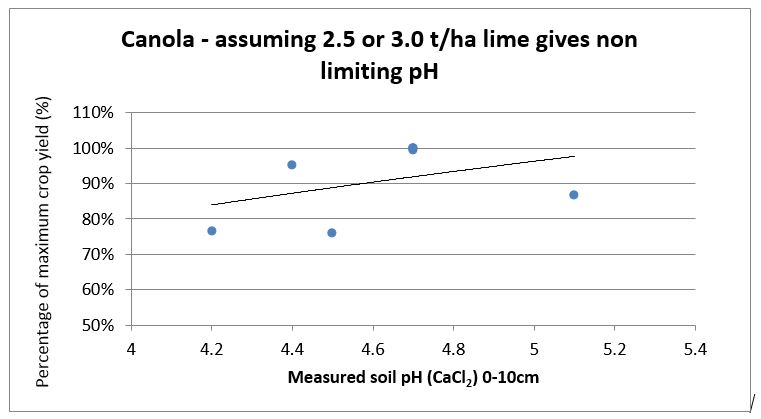
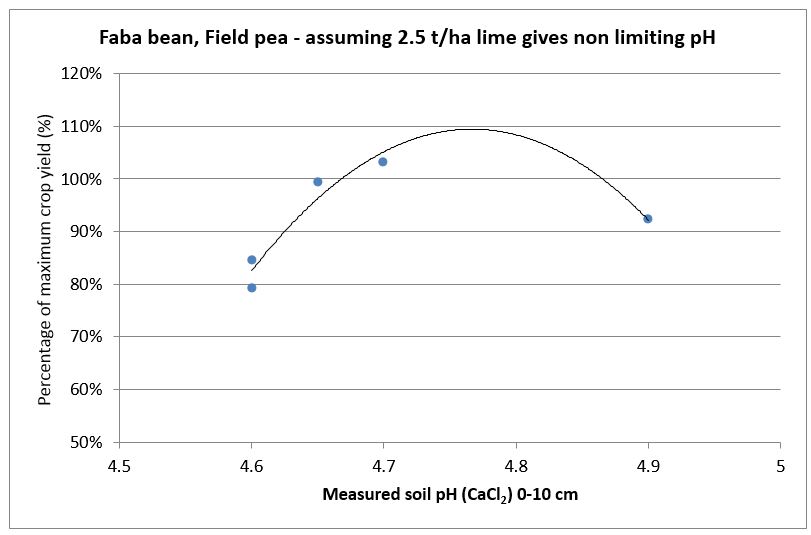
SFS replicated research trial data has been used to create lime response curves by calculating the percentage difference in yield of control un-limed plots compared to limed plots for wheat, barley canola and faba bean (see Figures 1-4). They show the yield reduction at different soil pH levels, especially in barley. The responses are from surface applied South West soft rock lime [Neutralising Value (NV) 90%, Effective Neutralising Value (ENV) 63%] in 2014 with minimal tillage and incorporation. Most of the trial sites had acidity less than pH 4.5 in the top soil plus acidity issues down to 15cm or 20cm. The moderate rates of lime applied and without any significant incorporation did not correct subsurface acidity at the sites and this probably reduced yield beyond what was measured.
Figure 1. Wheat yield responses to different soil pH levels measured 2012-2018 (n=17).
Figure 2. Barley yield responses to different soil pH levels measured 2014 to 2018 (n=6).
Figure 3. Canola yield responses to different soil pH levels measured 2012-2016 (n =7).
Figure 4. Faba bean and field pea yield responses to different soil pH levels measured 2014-2016 (n =5).
Lime responses are difficult to predict
The lime responses can be variable as they are influenced by many factors such as:
Subsoil acidity
A trial at Rokewood is investigating subsoil acidity further including incorporating lime and organic amendment (lucerne pellets) to depth (see Table 2). The pH at this site was 5.1 at 0-10cm, 4.1 at 10-20cm and 4.7 at 20-30cm. Relatively acid tolerant lupins were planted in the first year, but nonetheless it was apparent that the subsoil acidity resulted in a 672kg/ha reduction in biomass and a 200 kg/ha yield loss (although yield difference was not significant). Interesting the best response was with deep placement of lucerne pellets, producing an extra 399kg/ha biomass and 360kg/ha yield. Approximately a 20% significant yield reduction in both wheat and barley was recorded at an acidity trial site near Cootamundra, NSW where the acidity constraint was at 10cm-30cm depth.
Table 2. Rokewood subsoil acidity trial. Lupin response to liming treatments applied in 2018.
Treatments | Treatment description | Lupin yield (t/ha) | Establishment counts (Plants/m2) | Dry matter cuts at anthesis kg DM/ha | |||
|---|---|---|---|---|---|---|---|
Surface lime Incorporated | Surface liming 1.5t/ha incorporated into 0-10cm with offset discs to bring pH to 5.5. | 1.24 | b | 8.63 | a | 4084 | b |
Deep rip only | Surface liming 1t/ha incorp. + deep rip. Ripped down to 30cm, tines 50cm apart. | 1.36 | b | 6.73 | b | 4724 | a |
Deep lime | Surface liming 1t/ha incorp. + deep rip + deep lime. Deep lime 1.5t/ha placed between 8cm to 25cm (acid layer). | 1.44 | ab | 9.07 | a | 4756 | a |
Lucerne pellet | Surface liming 1t/ha incorp. and lucerne pellets 7.5t/ha placed into acid band. (Rate based on providing the same amount of alkalinity). Contains 200kg to 300kg N/ha. | 1.60 | a | 8.3 | a | 4983 | a |
LSD (P<0.05) | 0.21 | 1.43 | 537 | ||||
CV (%) | 7.46 | 8.72 | 5.8 |
LSD- Least Significant Difference, CV – Coefficient of Variation
Season
Acidity and aluminium toxicity reduce root growth which reduces the plant’s ability to extract soil moisture particularly in a dry finish to the season. However, if there is ample soil moisture and the crop’s nutritional needs are met, there may not be a significant lime response.
Organic carbon
High soil organic carbon (organic C >2%) appears to influence lime response by reducing the impact of aluminium solubility and toxicity.
Other soil constraints
If there are other soil constraints to production present, then a lime response may not be seen. This has been apparent in pasture trials in Southern Victoria where nutrient deficiencies were seen to over-ride lime responses (Miller, 2016). Lime or fertiliser may not have shown significant differences when applied separately, but together they did. This indicates that lime is not a substitute for fertiliser — both are needed. Other potential constraints may include compaction, waterlogging, or sodicity.
Lime reaction
Growers or agronomists looking for obvious responses in the first season following lime applications may be disappointed. Among the trials of this project, there were only two with statistically significant lime responses in grain yield in the first year and that was when pH was low (pH 4.2 and 4.5) and acid sensitive crops were grown. Lime needs acidity moisture and time to dissolve. Incorporated superfine lime has taken up to 18 months to fully dissolve. Surface applied lime without incorporation is likely to drive up soil pH in the top 1cm to 6.0 or above where lime stops dissolving. Hence, the benefit of incorporation of some kind.
Acidification rates
If you are farming and removing plant and/or animal products from the paddock then you are acidifying the soil. Soil acidification is caused by a number of processes, for example, as roots take up cations they release hydrogen ions to maintain charge balance. Also, the cycling of nitrogen is particularly important, with the addition of urea or ammonium which is converted into nitrate and then leached beyond the rooting zone, leaving behind acidity. If the agricultural system was closed (i.e. products not removed and nitrate not leached), then acidification rates would be zero.
Analysis of trial results and monitoring of 100 un-limed paddocks mainly within the Corangamite catchment showed that the rate of acidification varies according to the farming system and soil type (Table 3). The measured pH changes varied from 0.05 and 0.18 units per year depending upon the production system. The decrease in soil pH at 10-20cm and at 20-30cm over the four-year period was also found to be highly significant (p<0.05). The equivalent amount of pure lime (i.e. 100% NV) required to neutralise the annual acidification rate has been calculated from the reductions in pH over a four-year period (2014 – 2018) and is expressed as kg lime/ha/year. It was estimated up to 755kg/ha of pure lime p.a. is required to neutralise acidity from hay production on loamy soils throughout the soil profile.
Table 3. Average annual acidification rate measured across Corangamite farming systems at different depths based on pH (CaCl2) change over four years (2014 – 2018)
System | Average annual acidification rate of the soil layers (*Application of kg pure lime/ha/year to counteract acidity) | ||
|---|---|---|---|
0-10cm | 10-20cm | 20-30cm | |
Cropping on clay loams | Average pH fall 0.05/year Lime equivalent 180 (Range 85-430) | Average pH fall 0.03/year Lime equivalent 100 | Average pH fall 0.03/year Lime equivalent 100 |
Grazing on clay loams | Average pH fall 0.04/year Lime equivalent 138 (Range 85-345) | Average pH fall 0.01/year Lime equivalent 45 | Average pH fall 0.02/year Lime equivalent 50 |
Hay cutting including lucerne on loams | Average pH fall 0.18/year Lime equivalent 350 (Range 300-400) | Average pH fall 0.09/year Lime equivalent 175 | Average pH fall 0.12/year Lime equivalent 230 |
* Assuming 1 tonne of pure lime/ha lifts pH by 0.29 units in a clay loam and 0.5 in a loam.
Table 4. Acidifying effects of various farm enterprises in >500mm rainfall zone. (adapted from Hollier, 1999)
System | kg of lime/ha/year to balance acidification |
|---|---|
Continuous grain cropping including grain legume | 200 to 300 |
Grazed pastures | 100 to 200 |
Lucerne hay | 200 to 700 |
Lime movement takes time
When lime dissolves it produces bicarbonate ions (alkalinity) which neutralises hydrogen ions (acidity). Bicarbonate ions move 1cm to 3cm per year, depending on soil pH buffering capacity and leaching rainfall. In South West Victoria, after three to four years of applying lime with minimal disturbance in crop and pastures, the bulk of pH change was at 0-5cm and little change below it, except on light textured soil types in high rainfall areas. In crop trials, the average pH change was 0.2 below 5cm, although no pH change was measured at two trials. Norton et al (2018) also reported that after 11 years there was no lime movement beyond 5cm in a permanent pasture research trial at Sutton, NSW. The millennial drought may have contributed to the lack of movement, as well as the high pH buffering capacity of this clay soil.
Given the same rate of acidification, light textured soils (sands) show greater rates of soil pH decline than heavy clays because they do not buffer against the pH change as much (i.e. sands have a low pH buffering capacity). While the drop in pH due to soil acidification is greater on sands than clays, the pH increase following lime application is also greater.
Lime movement occurs if pH is kept above 5.5
Very few of the recent SFS trials saw lime movement beyond 10cm because there was not enough time for it to move and because rates were not high enough to saturate the surface with alkalinity to allow it to leach. Any change in pH measured may have been through physical lime falling down cracks. A lime trial run by NSW DPI from 1992 to 2010 (18 years) is one of a number of trials that only found subsoil amelioration when soil pH in the topsoil was kept above 5.5 (Li, et al. 2019). Micro-fine lime was incorporated into the top 10cm and soil pH maintained above 5.5 for the trial duration to counteract acidification and leaching. After four years, lime had moved to 15cm, but advanced no further for another four years (2004), but in 2010, lime was detected at 25 and 30cm. Movement was about 1cm per year.
Variation across the paddock and down the profile
The average paddock pH can be misleading when trying to make decisions about liming. To make informed decisions about liming, it is good to know what you are dealing with. The use of pH mapping or using yield maps to identify low production zones and then taking exploratory cores within them both have merit. The Rokewood subsoil acidity site provides a good example of how soil acidity changes spatially and vertically down the soil profile. The Rokewood trial is 100 by 140m and the variation in soil pH is 2 units in the top 10cm (Table 5).
Table 5. Average pH (CaCl2) and Exchangeable Al results for the Rokewood trial site 19/12/2017.
Depth | Average Soil pH | Range of soil pH | Average Al% of exchangeable cations |
|---|---|---|---|
0-10cm | 5.11 | 4.1-6.1 | 2.75% |
10-20cm | 4.10 | 3.8-4.4 | 19.33% |
20-30cm | 4.71 | 4.1-5.5 | 3.41% |
30-40cm | 5.76 | 4.8-7.1 | 0.09% |
Doug Crawford, Ag Vic, describes four considerations in relation to variable rate liming once the variability in soil pH is known. These are:
- No pH issues and therefore there is no need to lime.
- Marginal soil pH but maintenance liming is needed which can be applied by a blanket rate.
- There are distinct pH zones which make variable rate useful.
- There is too much variability but generally low pH, therefore it makes sense to apply a blanket rate of lime.
Doug describes pH mapping as an insurance policy to make sure lime is applied to where it is needed most and hence, is as cost-effective as possible. Some argue that the money spent on pH mapping (approx. $65/ha) is better spent on additional lime. Having pH maps allows you to identify zones that can be monitored in future, especially if subsoil acidity is likely.
Variation in soil pH across paddocks is caused by management and soil type which makes it hard to predict. Some examples of variation are:
- Sheep stock camps (see McCaskill, 2009, who showed how sheep camps affected the establishment of lucerne at the Hamilton Evergraze Proof site).
- Lime spreading inaccuracies (see Burns and Norton, 2018, for examples of lime misses in faba bean).
- Burnt canola swaths increased soil pH by 0.3-0.8 of a pH (CaCl2) unit, equivalent to about 1t/ha of lime in DAFWA investigations (Brennan, et al. 2003).
- Trees generally increase soil pH, but blue gums are an exception.
- High production areas tend to have more product removal resulting in more acidity.
- Cutting hay of half or parts of the paddock. Hay cutting is highly acidifying.
Developing a lime program
Once the distribution of soil acidity is spatially and vertically understood, decisions can be made about where to lime, what rates are needed and how to apply the lime. Most growers and advisers only consider the 0-10cm soil. Lime rates are rarely estimated based on treating acidity at all soil depths — 0-10cm, 10-20cm and 20-30 cm, which is why subsoil acidity develops.
Lime rates are determined by knowledge of the pH buffering capacity, which is chiefly influenced by the amount of organic carbon and then clay content. Below is a commonly used method for lime rate estimation which is available through Soil acidity monitoring tools (DPI, 2005) and appears in OptLime and Soil Amelioration calculators from WA. It calculates the amount of lime required to reach a target pH but to maintain it.
The target soil pH for the 0-10cm is 5.5 to 6.0, if aiming to achieve lime movement or growing acid sensitive pulses, and pH 4.8 deeper in the soil to avoid Al toxicity.
Step 1. Pure lime requirement (t/ha) = (Target pH – Current pH) ÷ Conversion factor
Divide the desired pH change by a conversion factor for different soil types:
0.26 for clay,
0.37 for clay loam,
0.47 for sandy clay loam,
0.57 for sandy loam and
0.67 for sand.
Step 2. Adjustment for organic matter (OM) - If the soil OM% is above 2%, then add an extra 0.4t/ha.
Note OM% = organic carbon% x 1.7. Greater than 2% OM content is likely in most pastures or crop pasture rotations.
Note the calculation is for pure lime which has 100% NV and so lime rates will require adjustment depending on the NV% of the lime to be used.
Also, these calculations need repeating for each 10cm soil layer as shown in the example in Table 6. The 10-20cm and the 20-30cm layer will be unlikely to contain OM content below 2% so step 2 can be ignored.
Table 6. The calculations of approximate lime rate required at the Rokewood sub soil acidity trial.
Depth | Average Soil pH | Target | pH changed required | Soil type | Organic C% | Lime requirement (t/ha) |
|---|---|---|---|---|---|---|
0-10cm | 5.11 | 5.8 | 0.7 | Sandy loam | 0.8 | 1.2 |
10-20cm | 4.10 | 4.8 | 0.7 | Sandy loam | 0.5 | 1.2 |
20-30cm | 4.71 | 4.8 | 0.1 | Clay | 0.6 | 0.3 |
Total pure lime requirement for soil pH recovery | 2.8 | |||||
Lime calculation rates for amelioration of acidity are estimates only and so it is important to monitor soil pH change after three or five years so that rates can be adjusted.
Too much lime?
Topdressing large amounts of lime without incorporation may cause micronutrient deficiencies (e.g. copper, zinc, boron and manganese) if these elements are already marginal, especially on poorly buffered sands. High rates of pure lime (NV 100%) are thought to be in excess of 2t/ha for a sand, 3t/ha for a sandy loam or 4t/ha for a loam or clay loam soil. While these deficiencies can be overcome by the application of appropriate granular fertiliser or foliar sprays, if high rates of lime are required, it is best to split applications over a period of three or four years or incorporate the lime with a tine or disc implement.
Choosing a lime
Choosing a lime should be based on the most cost-effective product. Transport costs can be high so often, the cheapest lime is from the pits located closest to the farm. The price of lime tends to reflect the level of processing. However, you need to know how effective the product is at neutralising acidity.
The typical costs of lime spread are about $40 to $50/t depending upon the quality and transport distance.
- Purchase cost $18 to $30/t for Victorian Ag lime.
- Cartage rate 10 cents/km.
- Spreading $8/t.
- Variable rate spreading, extra $2/ha.
Most agricultural limestones are calcium carbonate and can be described as hard rock limes or soft rock limes. Explosives are needed to extract hard rock limes and need to be processed very finely so they react quickly in the soil, which add to the expense. Most of the limes in Victoria are soft rock excavated, crushed and then particles screened to less than 2mm. One of the best quality limes is from near Goulburn, NSW, which is 99% purity and ball milled so that 70% of particles are less than 0.01mm, costing approx. $39/t. However, if it must be transported 600km, it is not cost effective.
Purity, particle size and solubility
The main factors determining lime quality include purity (i.e. neutralising value NV) particle size distribution and solubility (Scott et al. 1992). Pure calcium carbonate (or pure limestone) has an NV of 100%. The higher the NV, the purer the product. Lime products sold in Victoria commonly have an NV of 80 to 90%. Some products can exceed 100% if containing appreciable amounts of magnesium carbonate and/or burnt lime.
The finer a lime product is, the greater the surface area for the neutralising chemical reactions to occur. Therefore, a finer lime will reduce soil acidity more quickly than a coarser lime. The rate of lime dissolution is also affected by its solubility. Lime is regarded as being relatively insoluble, although this varies amongst different types. NSW DPI compared the pH change of different lime types with the same average particle sizes and found that soft limes created 20% greater pH change compared to a hard calcitic lime over a period of six to 12 months due to differences in solubility (Conyers et al. 1995). Dolomites contain less soluble magnesium carbonate, and created 15% less pH change compared to hard limes. There are no standard tests for lime solubility.
Effective Neutralising Value (ENV)
ENV is a calculation that allows for comparison of different liming materials by accounting for both the neutralising value of the lime and particle size. An ENV of a lime product is calculated based on the sum of its percentage of particle sizes (fine <0.3mm, medium >0.3mm to 0.85mm and coarse >0.85mm) which are discounted according to their potential reactiveness. Particle sizes of 5mm are thought to take about 20 years to dissolve. This indicated a flaw in the ENV quality assessment as there is a vast quality difference in limes with particle sizes 1mm to 2mm and those with greater than 5mm which is currently not accounted for.
The Victorian Lime Producers Association (VLPA) members annually report NV and particle sizes greater than 1mm, which is made available on their website. A good quality lime from Victoria will have close to 50% of its particle sizes less than 0.3mm and very little above 0.85mm and with good screening practices should contain no more than 5% above 1mm. Having up to date information on particle size distribution is important, even for soft rock limes. Independent tests are available from laboratories such as Agrifood Technology, CSBP or Apal. There are several calculators available to help compare limes based on their NV% or ENV, such as Lime Cheque, available off the Ag Excellence Alliance website (https://agex.org.au/project/soil-acidity/).
Pelleted limes and liquid limes
There has been interest in the use of pellets containing super fine lime (20 to 40 µm) for ameliorating soil acidity, however, the costs of these products are about $260/t in bulk. Researchers in WA have reported that the lime pellets used in their trials acted like a good quality lime, and that it did not move horizontally in soils as was hoped, only vertically. There is interest in using these products in air seeders to place prilled lime into acidic layers at 10-20cm at rates of 500kg/ha for two or three years and possibly in different orientations to maximise soil coverage.
Liquid lime sources are generally micro-fine calcium carbonate in suspension. In trials conducted by the Woady Yaloak catchment group, recommended rates of liquid limes were shown to be ineffective at creating pH change in comparison to standard rates of agricultural limes. Even though liquid limes have high NV the recommended rates are insufficient to correct soil acidification rates. It has been suggested that a liquid lime solution might wash through the soil macro-pores and may correct acidity at depth, but this has not been demonstrated in field trials.
Profitability of liming
The profitability of liming is generally straightforward because many of the crops are acid sensitive, such as barley and pulses, and liming costs are often recouped within short time frames (see Li et al. 2010). Certainly, at one SFS trial, acidity cost was calculated to be $734/ha over the three-year period and liming (approx. $150/ha spread) paid for itself after the first year (see Table 7). This trial provides a good example of how liming can avoid yield losses from acidification.
Table 7. Costs of soil acidity at the Bellarine trial site where soil pH was 4.2 (0-10cm), 4.4 (10-20cm) and 4.9 (20-30cm) and lime applied at 3t/ha.
Year | Crop | Average yield of limed plots (t/ha) | Yield reduction compared to lime 3t/ha | $Price/t of grain | $ Calculated cost of acidity |
|---|---|---|---|---|---|
2014 | Barley | 3.0 | 1.0 | $278 | $275 |
2015 | Canola | 1.7 | 0.4 | $531 | $228 |
2016 | Wheat | 7.3 | 1.2 | $200 | $230 |
Total costs of acidity for 3 years | $734/ha | ||||
Timing of lime application
Lime can be applied any time. Lime is normally applied in summer or early autumn to fit the farming schedule. Spring liming of pastures is beneficial to a following cropping rotation as there is time for the lime to start neutralising acidity, particularly if there is summer rainfall. The ‘Liming acidic soils for legumes’ project advises that applying liming preferably two years before a sensitive crop such as pulses.
The factors which affect the time of liming are:
- Paddock trafficability.
- The need to apply lime six weeks prior to sowing to avoid inducing any micronutrient issues.
- Accessing lime, and ability to back-load lime after grain delivery to reduce transport costs.
- Wind erosion risks where the fine component of lime is lost from bare surfaces.
Growers in high rainfall zones often look to spread lime prior to burning stubble. Lime effectiveness is not adversely affected by burning. Agricultural limes are treated at 900oC in kilns to create burnt lime which makes it faster acting, for use in the building and horticultural industries. A hot stubble burn might reach 500oC at most. However, agronomists raise some concerns with spreading lime before burning stubble which need to be considered. These include:
- Spreading lime over heavy stubble may not give even coverage compared to spreading on bare paddocks. This may be a visual perception issue as the lime cannot be as easily seen amongst the stubble or could be valid if the spreading height was too low.
- Stubble does not seem to burn as well if lime is spread. This may be a factor if the lime was moist and covered stubble.
Applying lime into paddocks with some stubble cover or prior to burning is likely to reduce losses from wind erosion. Timing application to coincide with rainfall or heavy dews can help wash the lime off any stubble and start to dissolve the lime.
Conclusion
Acidification occurs in all farming systems where plant or animal products are removed, and liming will be necessary to neutralise it or else significant losses in yields will occur
Acidification is not just confined to the top 10cm of soil and the emerging issue of acidity at 10-20cm indicates that our liming practices (rates, frequencies and lack of incorporation) have been insufficient at addressing it. Therefore attention to the subsurface and subsoil through monitoring, the maintenance of good soil pH and generally incorporation of lime in low rainfall (<500 mm) or heavy clay soils is vital for protecting the soil asset.
Useful resources
http://www.sfs.org.au/SoilAcidityLimeResponse
https://agex.org.au/project/soil-acidity/
https://grdc.com.au/legumes-in-acidic-soils
https://www.agric.wa.gov.au/soil-acidity/soil-acidity-frequently-asked-questions-faqs
http://vlpa.asn.au/w/product-specification/
References
Brennan RF, Bolland MDA and Bowden JW (2003). Post canola syndrome: burning swathed canola stubbles enables potassium deficiency and induced molybdenum deficiency and aluminium toxicity to be expressed in the next cereal crop. In ’Thirteenth Biennial Australian Research Assembly on Brassicas’ (J Edwards, ed.), pp. 32-39. NSW Agriculture, Orange Australia.
Burns H and Norton M (2018). Legumes in Acidic Soils maximising production potential in south eastern Australia. ISBN 978-1-921779-71-8 (Online) Grains Research and Development Corporation and NSW Department of Industry.
Conyers MK, Scott BJ, Fisher R and Lill W (1995). Predicting the field performance of twelve commercial liming materials from southern Australia. Fertiliser Research 44, 151-161.
DPI (2005). Soil acidity monitoring tools. ISBN 1 74146 533 8. Victorian government Department of Primary Industries.
Elliot M (2009). Grass tetany in cattle. NSW DPI Prime Fact 420. NSW Department of Primary Industries.
Fenton G (2003). Planning on liming, NSW Agriculture, Acid Soil Action leaflet No 4 (2 ed).
Hollier C. (1999). In “Acid Soil Action.” Land and Water Resources Research & Development Corporation.
Hollier (1999). Acid Soil Action: investment for your soil now and in the future. pp 34. DNRE, Victorian Government.
Li GD, Singh RP, Brennan JP and Heylar KR (2010). A financial analysis of lime application in a long-term agronomic experiment on the south-western slopes of New South Wales. Crop and pasture science 61, 12-23.
Li GD, Conyers MK, Heylar KR, Lisle CJ, Poile GJ and Cullis BR (2019). Long-term surface application of lime ameilorates subsurface soil acidity in the mixed farming zone of south-eastern Australia. Geoderma 338, 236-246.
McCaskill MR, Saul G, Clark S, Cameron F, O’Brien, Behrendt R (2009). Evergraze: establishment of lucerne at the Hamilton proof site. In ’Proceedings of the 50th Annual conference of the Grassland Society of Southern Australia’.
Miller L (2016). What responses can I expect from liming pastures. In ‘Trial Results Victoria Edition 2016’. pp 112-116 Southern Farming Systems.
Norton MR, Garden DL, Orchard BA and Armstrong P (2018). Ameliorating acidity of an extensively- managed permanent pasture soil. Soil use and management 34, 343-353.
Paige KL, Dalal RC, Dang YP, Kopittke PM, Kirchof G, Fujinuma R and McKenzie’s NW (2018). Management of the major chemical soil constraints affecting yields in the grain growing region of Queensland and New South Wales, Australia – a review. Soil Research 56, 765-779.
Scott B J, Conyers M K, Fisher R and Lill W (1992). Particle size determines the efficiency of calcitic limestone in amending acidic soil. Australian Journal of Agriculture Research 43, 1175-1185.
Scott BJ and Conyers MK (1995). Magnesium nutrition and lime movement down the profile. In Making better fertiliser, lime and gypsum recommendations. In Proceedings of a workshop, Agricultural Research Institute, Wagga Wagga. (Eds IG Fenton and PW Orchard) pp. 1-18. NSW Agriculture.
Acknowledgements
The research undertaken as part of this project (SFS00026)is made possible by the significant contributions of growers through both trial cooperation and the support of the GRDC — SFS would like to thank them for their continued support. Other acknowledgments include the Australian government support through the Corangamite catchment management authority for pasture lime response trials and soil benchmarking and through a Smart Farms grant ‘Building Farming Resilience’. In addition, thank you to NSW Department of Primary Industries, ‘Managing Subsoil Acidity’ project and Agriculture Victoria, ‘Spatial Variability of Soil Acidity & Liming’ Project.
Contact details
Lisa Miller
Southern Farming Systems
23 High St, Inverleigh, Vic 3321
0488 600 226
lmiller@sfs.org.au
GRDC Project Code: SFS1401-001RTX, DPI1501-003RTX, SFS1811-001OPX,