Within-paddock nitrogen variability and the potential role of cereal grain protein mapping for site-specific N management
Author: Eva Moffitt (EM Ag Consulting) | Date: 14 Jul 2021
Take home messages
- Wheat grain protein concentrations of less than 11.5 % generally indicate that nitrogen (N) supply was insufficient for a crop to meet its water limited yield potential
- If this ‘rule-of-thumb’ is applied across a landscape, a spatially referenced wheat grain protein concentration map is analogous with an ‘N adequacy’ map
- This layer can be used in conjunction with targeted deep N soil sampling as a basis for site-specific N inputs to reduce both instances of yield loss due to N undersupply and adverse environmental/economic consequences associated with N oversupply
- Research conducted in 2019/2020 across five paddocks (511.4 ha) in southern NSW supported the use of wheat protein mapping as a basis for site-specific N
- This approach may have a good fit for growers located on soils not prone to N losses that have variability in factors such as texture/CEC/OC%/PAWC, productivity (N removal) and/or management histories (e.g., amalgamated paddocks, variable N inputs)
Introduction
This paper will explore concepts around managing nitrogen (N) variability as part of a larger (whole system) approach to improving N management, with particular emphasis on the potential role of cereal grain protein mapping in site-specific N fertilisation. Results will be presented from paddock scale research conducted in 2019-20 that examined relationships between soil mineral nitrogen (SMN) levels and grain protein concentration across five paddocks in southern/central NSW.
The broader problem
Despite widespread knowledge of the importance of nitrogen (N) supply in broadacre cropping systems, N deficiency remains the most substantial contributor to the sizeable yield gap in Australian wheat production (Hochman and Horan, 2018). Furthermore, recent assessments have found that most Australian grain cropping systems are in negative N balance, that is, more N is being exported off-farm than is being applied as fertiliser or fixed from atmospheric N2 (Angus and Grace 2017).
A major driver of N under-supply has been the naturally conservative approach of growers operating in highly variable rainfall environments, where adverse agronomic and environmental consequences of N oversupply have been experienced and/or are perceived. To this end, Australian growers have historically placed much reliance on mineralisation of organic N to meet crop demands (Angus et al., 2006), which has been desirable not only from an economic perspective, but also agronomically due to the positive relationship between both N supply and crop demand with soil moisture.
With resultant declining levels of soil organic matter (SOM; up to 60% under continuous cropping; Dalal & Chan, 2001) and the diminishing adaptive N supply ability of our soils, it is clear that a growing requirement exists for fertiliser N to maintain (or increase) productivity within the Australian grains industry.
For fertiliser rates to rise in a sustainable manner, it is also clear that there is considerable work to be done in improving the robustness of N fertiliser decision methods. In a 2015 survey of 132 commercial crop advisors in New South Wales, Schwenke et al. (2018) found that while most advisors regarded soil tests as moderately to very important for determining N fertiliser requirements, interviewed participants revealed that many of their clients either did not soil test, or of those that did, the number of paddocks tested was quite low. This supports findings by Lobry de Bruyn & Andrews (2016), who found that only 25-30% of Australian broadacre crop businesses conduct annual soil testing for nutrient levels.
One of the key barriers to N soil testing identified by Schwenke et al. (2018) is the view among growers that within-paddock spatial variability of N is high, which leads to distrust of whole-paddock bulked soil test results. Growers are instead more comfortable using ‘rules-of-thumb’ approaches based on paddock history and seasonal outlook. This suggests the lack of cost-effective, sound agronomic methods for quantifying and mapping spatial N variability is a substantial impediment to the overall improvement of N management in Australian cropping systems. This is supported by on-the-ground experience which would suggest that the lack of trusted variable rate (VR) N solutions available to growers presents a far greater impediment to the adoption of precision N practices than technological capacity or grower enthusiasm.
Supply and demand concepts
To better understand the challenges of successful site-specific N approaches, it is useful to examine the basic N dynamics at play in broadacre cropping systems. In simple terms, optimal N management refers to matching N supply to N demand – both parameters of which can be highly spatially variable in the Australian landscape.
For example, on the supply side of the equation – residual (carryover) N may vary according to previous crop and pasture productivity (influencing both N removal and N fixation), in-season mineralisation may vary according to soil type and management history (influencing SOM pools and moisture), N losses may vary according to factors such as soil texture and/or landscape position (influencing leaching and waterlogging/denitrification) while a myriad of other less predictable factors and/or interactions may also be at play (e.g. uneven fertiliser/manure applications, uneven removal of hay, redistribution of N by livestock).
On the demand side of the equation, variability of yield potential in the Australian landscape can be substantial over very short distances, often driven by differences in plant available water-holding capacity (PAWC) resulting from variability of soil properties such as texture, bulk density and subsoil constraints (Rab et al., 2009).
This presents a highly complex situation where both supply and demand of N may be spatially variable due to entirely different (and often independent) driving factors. To further complicate the situation, many of these factors are temporally variable, making it difficult to correctly quantify whole season patterns of N deficit using data collected at any one snapshot in time. For example, the spatial patterns of start-of-season SMN may not match those of the full season N supply if there are considerable differences in mineralisation between different zones of the paddock.
Current approaches to site-specific N in Australia
There are two main approaches to sub-paddock scale N management currently in practice and/or commercially available in Australia.
The first is to divide a paddock into a number of sub-units or ‘management zones’, which are considered more-or-less homogenous in their N supply and/or demand attributes (Rab et al., 2009). Zones are generally developed based on either historic productivity (e.g., using yield and/or remotely sensed imagery) or soil type (e.g., using apparent electrical conductivity (ECa), grid soil Cation Exchange Capacity (CEC) mapping and/or aerial imagery). Each zone is then soil sampled separately and managed accordingly. While these approaches are generally considered to be an improvement on whole paddock testing, their main limitation is that the resolution of data collection is still quite low, therefore substantial reliance is placed on the accuracy of the zoning process. As moisture availability is generally the greatest yield constraining factor in Australian systems, it is likely that these methods do a reasonable job of differentiating areas of contrasting N demand, however, may not be as effective at detecting finer scale variability of N supply. To date, the majority of research around the accuracy of different zoning approaches has focused on crop responsiveness/N demand, with very little work assessing the homogeneity of N supply within zones (i.e., quantifying SMN variability).
The second approach is the use of remote or proximal sensing to directly develop site-specific N maps for mid-season N fertilisation. The most widely implemented spectral index used for this purpose is the Normalised Difference Vegetation Index (NDVI), which gives a representation of the amount of photosynthetically active biomass in a crop (Perry et al., 2014). These approaches have similar limitations to productivity-based management zone methods in that mid-season biomass is often correlated more closely with moisture availability (or other factors) than N nutrition per se. To address these limitations, work continues to identify alternative spectral indices that are more directly related to N nutritional status (e.g., CCCI, Basso et al., 2016; Red Edge, Richetti et al., 2020). In either case however, the successful implementation of these strategies generally requires the use of N-rich and N-poor calibration strips, ground-truthing and a good understanding of site-specific yield potentials relative to seasonal conditions. Indeed, a recent review by Colaço and Bramley (2018) of a large suite of globally published sensor evaluation studies found a lack of consistent evidence to confirm whether crop sensors in isolation can deliver benefits to N management. Instead, they suggest that future success will come in the way of more sophisticated algorithms that integrate spectral data with input from other sensors and data layers (e.g., moisture probes, weather forecasts, ECa mapping, etc.).
In addition to these two primary methods, there has also been limited use of 2-4 ha resolution grid deep N soil mapping for site-specific N in Australia, however this approach has generally been considered uneconomical due to the relatively expensive nature of deep sampling (Bramley and Janik, 2005).
Theoretical background to cereal grain protein based site-specific N
For many decades it has been recognised that a consistent relationship exists between cereal grain yield and cereal grain protein concentration according to N supply (e.g., Russell, 1963). This relationship consists of increasing grain yield and protein concentrations with greater N supply up to a certain point, after which grain yield begins to plateau while protein concentration continues to increase. At very high N levels, a decline in yield often occurs (Holford et al., 1992).
The point at which N supply has been optimised for maximum grain yield is termed the ‘critical grain protein concentration’ and has been found to be around 11.2–12.0% in most Australian hard white wheats through studies conducted in southern/central NSW (Brill et al., 2013, Sandral et al., 2018) and South Australia/Victoria (G. McDonald, review published in Unkovich et al., 2020).
While critical grain protein concentrations will vary between varieties and across seasonal conditions (Fowler, 2003), a simplified ‘rule-of-thumb’ interpretation under favourable (non-drought) conditions can be summarised as:
- Protein < 11.5% = insufficient N supply to meet yield potential
- Protein 11.5–12.5% = adequate/optimum N supply to achieve yield potential
- Protein > 12.5% = surplus N to crop requirement, possibly some yield penalty (Figure 1).
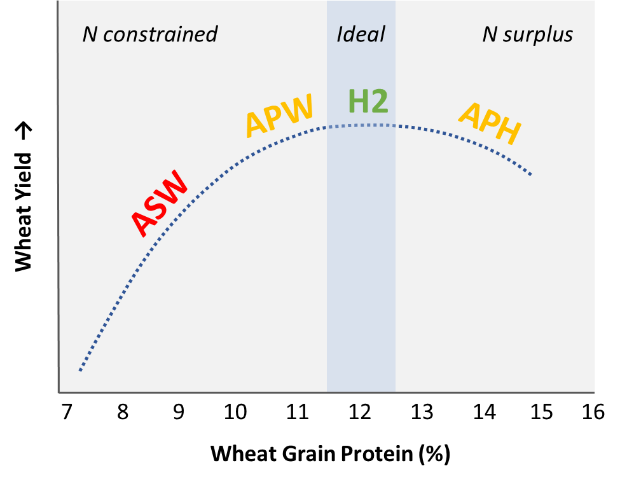
Figure 1. A generalised representation of the relationship between yield and grain protein concentration in wheat with increasing N supply. Labels refer to grades in the Australian wheat classification system
If we apply this rule-of-thumb spatially across a management area grown to a single wheat variety, a georeferenced map of wheat protein concentration is analogous to an ‘N adequacy’ map – i.e., it serves to distinguish areas of the paddock that had insufficient, ideal or surplus N according to their site-specific yield potentials. Provided that crop demand wasn’t very much higher than budgeted N supply (resulting in a full drawdown of soil mineral nitrogen (SMN) across the paddock), and that soils are not prone to N losses, it is likely that protein patterns will provide a good spatial representation of residual (carryover) SMN.
Ground-truth soil testing at the start of the following season can be used to test this assumption and quantify out-of-season mineralisation. A good approach to determining the placement of soil tests is to divide the paddock into zones based on combinations of yield and protein results from the previous harvest. This process provides useful insights into not only N dynamics but also where non-N related constraints may warrant further investigation. These concepts are summarised in Table 1.
Table 1. Within-paddock combinations of cereal yield, protein attributes and their properties.
Classification | Interpretation | Residual N levels | Action |
|---|---|---|---|
High Yield / | - Optimum scenario - This area of the paddock may have higher mineralisation potential | Likely moderate to high, however soil test to confirm (particularly if crop N demand was higher than budgeted) | Determine rates based on soil test results and according to high yield potential |
High Yield / | - Sub-optimal N management | Likely low | Increase N rates relative to paddock average in following season/s to support higher yields and build SMN |
Low Yield / | - Non-N related problem | Likely high | If the constraint cannot be amended, reduce N inputs relative to paddock average permanently to match lower yield potentials |
Low Yield / | - Sub-optimal N management | Likely low | Start by increasing N to determine the non-N constrained yield potential, then manage according to results |
A major advantage of a protein-based VR N approach over currently available alternatives is that it combines both the supply and demand elements of the N balance equation. For example, low protein areas within a paddock may occur either due to low N supply (i.e., differences in carryover N, mineralisation, fertiliser inputs, etc.) OR higher yield potential (i.e., due to the dilution of protein by higher yield; Simmonds, 1995). Regardless of which factor is responsible (or both), the management decision will involve increasing N rates in the following season.
In this sense, the protein layer is also accounting for temporal variability of N dynamics by providing a retrospective assessment of the whole season, net N balance, rather than a ‘snapshot in time’ as occurs with data layers such as spectral indices or grid soil mapping.
Another advantage is the benefit afforded by the plant providing an indication of N adequacy according to the conditions it experienced, i.e., the plant available N. This circumvents a limitation of soil testing where mineral N may be present within the profile however the plant may not be able to access it (e.g., if subsoil hostilities prevent root access). In a similar manner, if subsoil conditions are favourable and the plant is able to access deeper SMN, this will be reflected by the plant’s protein concentration however may be missed by an arbitrary soil sampling depth cut-off.
Setting rates
Due to fluctuations that occur in critical grain protein concentrations between seasons and some varieties, start-of-season soil sampling will remain an essential step to determining actual N rates. Soil sampling will also act as a ground-truthing step to test assumptions regarding patterns of carryover SMN and to test any unusual areas (e.g., if losses are suspected such as where waterlogging has occurred).
Where consistent protein zones are present, soil sampling should cover off on each of the major protein/yield combinations (see Table 1), aiming to get an idea of the paddock average and the spread (range) of SMN values. If protein data across the paddock is spatially noisy or does not have consistent zones, the paddock may not be a good candidate for VR N (i.e. SMN may not vary substantially, or variability might be on a sub-manageable scale). In this case, whole paddock testing and blanket rates may be more appropriate.
Once a paddock average has been determined, growers and advisors can use their preferred calculation method or decision support system (e.g. Yield Prophet®, ‘N banks’) to determine a ‘base rate’ which will act as the paddock average from which to vary N inputs (i.e. lower rates on high protein areas, higher rates on low protein areas). The increments of difference between rates will depend on; a) the spread of protein values, b) the spread of soil test results, and c) the grower’s level of confidence/conservatism. A more conservative approach (smaller increments) will afford a lower level of risk when moving from blanket rate applications, however there will likely be a longer lead time in reducing within-paddock N variability.
Over a number of seasons, implementing this strategy should reduce the spatial variability of protein concentrations, ideally converging around 11.5 - 12.5% if the base rates chosen have been appropriate. It is likely that the most ‘bang for buck’ to be gained implementing this strategy will occur in the early stages, by eliminating very low (highly constrained) and very high N zones.
It is important to remember that in paddocks where yield potential varies greatly due to factors other than N (e.g., relatively fixed factors such as PAWC), a successful outcome will not be where yield becomes even, but rather where yield is optimised in all areas according to their site-specific yield potentials. In these instances, N rates will need to continue to be varied to match N supply with variable N demand. One option for achieving this may be to transition to a VR N strategy based on N removal patterns.
In all cases, ongoing monitoring of cereal protein% results and annual deep soil sampling should serve as a constant feedback to ensure N decision-making approaches are performing well.
Getting started
A protein based site-specific N strategy might be a good approach for a grower if they:
- Are predominantly located on soil types not prone to losses (i.e., free draining with good nutrient holding capacity such as occurs across most of southern NSW), and
- Have within-paddock variability in factors such as texture/CEC/OC%/PAWC, productivity (N removal) and/or management histories (e.g., amalgamated paddocks, previous inputs).
At present, the cost of a harvester mounted grain analyser is around AUD $25,000 + GST including installation (Next Instruments ‘CropScan 3300H’ unit). This cost will be spread over a number of seasons. The unit can also be removed and reinstalled if a new harvester is purchased. There will also be costs related to data management and interpretation if the grower cannot or does not wish to do this themselves.
After completing the first harvest, a good strategy is to pick a few of the most variable paddocks to focus on. If a grower isn’t comfortable implementing a VR application straight away, they may prefer to use N-rich and/or N-poor strips to test the impact of variable N rates on their soils. If doing so, strips should be designed so they pass through several zones (e.g., low/high protein, soil types, management histories, etc.). Paddocks being cropped to a second cereal crop (e.g., wheat on wheat) will be of most value for reviewing the results of strip trials and/or the success of VR N applications.
Research results
A 2019/2020 research project undertaken in southern NSW by FarmLink Research in conjunction with Precision Agriculture sought to examine within-paddock N variability patterns and test assumptions around the correlation of SMN with various parameters, including protein concentration. Selected findings are presented below. The full research report can be accessed at http://www.farmlink.com.au/project/nitrogen-variability (Moffitt, 2021).
Aims
- Quantify levels of within-paddock SMN variability across five cropping paddocks (511.4 ha)
- Examine correlations between 2020 start-of-season SMN and various other parameters including 2019 yield, protein and N removal, ECa (via EM38), soil texture and OC% (via MIR)
- Comment on the effectiveness of each layer to inform site-specific inputs, and
- Develop grower and advisor capacities for VR N decision making.
Methodology
Georeferenced yield and grain quality data was collected during harvest 2019 by eight late model Case IH harvesters equipped with standard yield monitors and retrofitted CropScan 3000H grain analysers. All CropScan 3000H units were calibrated prior to harvest using a single set of certified reference samples for wheat, barley and canola (protein%, moisture% ± oil%).
Five paddocks (four wheat, one barley) were subsequently selected on the basis of having complete yield/protein datasets and some level of protein variability. Paddocks were all located within 100 km of Temora in southern NSW on predominantly red to grey sandy loam to clay loam topsoils overlying clay loam to clay subsoils (chiefly Chromosols/Sodosols). Paddock management has consisted of continuous cropping of cereals (wheat/barley), canola and occasional pulses, with some paddocks having histories of lucerne/clover phases. Annual rainfall across the five sites averages around 480-600 mm however in 2019 rainfall was very low, ranging from 160-310 mm (annual) and 64-142 mm (April-October). As a result, none of the five paddocks had any additional N applied throughout the 2019 season apart from low levels in MAP/DAP fertilisers applied at seeding.
Grid soil sampling plans were designed at resolutions of 1.17 ha (108 m x 108x m; 4x paddocks) and 1.44 ha (140 m x 140 m; 1x paddock), depending on the width of top-dressing operations. A total area of 511.4 ha (425 grid sites) was soil sampled in late February/March 2020 at 0-30 cm/30-60 cm intervals and analysed for nitrate (NO3), ammonium (NH4), MIR texture and MIR Organic Carbon% (OC%). Soil sampling occurred after opening rainfall in 2020 following extremely dry conditions for many months prior. EM38 and elevation mapping was conducted during January 2020 at 18 m/24 m swaths. Weighted averages for each grid cell location were determined for EM38/elevation and yield/protein data through various interpolation methods. The strength of the relationship between SMN and other attributes was analysed via linear regression at the grid resolution.
Grower-led VR N applications and post-harvest grid soil mapping were also conducted in 2020. These results will not be discussed in detail below however can be obtained in the full report.
Results and discussion
Considerable within-paddock variability of start-of-season (Feb/Mar 2020) SMN was observed at four of the five sites, where the range of values (max – min) was greater than 140 kg N/ha, and the standard deviation was greater than 20 kg N/ha (Table 2). At the fifth site (Ardlethan), where the average SMN was much lower (46 kg N/ha ± 11 kg N/ha SD), the range of SMN was 43 kg N/ha.
Table 2. Summary of 2020 start-of-season 0-60 cm SMN and 2019 harvest protein% results
(grid n = 58 to 96)
Ardlethan (83 ha) | Girral (103 ha) | Rannock (103 ha) | Temora (111 ha) | Thuddungra (112 ha) | ||
|---|---|---|---|---|---|---|
Feb/Mar 2020 0-60 cm SMN (kg N/ha) | Mean | 46 | 94 | 95 | 67 | 127 |
Min | 27 | 29 | 52 | 22 | 55 | |
Max | 70 | 213 | 199 | 162 | 285 | |
SD | 11 | 40 | 24 | 26 | 38 | |
CV | 24% | 43% | 25% | 39% | 30% | |
2019 Protein % | Mean | 8.3 | 17.4 | 11.7 | 13.4 | 15.3 |
Min | 6.7 | 15.9 | 10.0 | 10.1 | 14.0 | |
Max | 11.9 | 18.4 | 13.5 | 15.4 | 16.8 | |
SD | 1.2 | 0.7 | 0.8 | 1.3 | 0.6 | |
CV | 14% | 4% | 7% | 10% | 4% | |
When examining the relationship between start-of-season (Feb/Mar 2020) SMN and various other attributes, 2019 grain protein% displayed the most consistent and strongest correlation compared to all other layers examined (Figure 2a; Table 3). This consistently positive relationship was significant at 4 out of 5 sites. The site that did not show a strong correlation (Ardlethan) was also the site of the lowest average SMN, lowest range of SMN and lowest average protein% levels (8.3%). Interestingly however, this site had one of the highest ranges of protein concentration (6.7% – 11.9%) in comparison to all others. This result will be discussed in more detail further below.
Figure 2. 0-60 cm Soil Mineral N (kg N/ha; sampled Feb-Mar 2020) versus 2019 cereal harvest results, (a) Grain Protein Concentration and (b) Dry Yield. (Girral = barley, rest = wheat). Each point represents one grid site (n = 425)
Table 3. Pearson correlation coefficients (r) for start-of-season (Feb/Mar 2020) 0-60 cm Soil Mineral N versus various attributes for each of the five trial paddocks. Values in bold are significant at
P < 0.0001. *Variable rate chicken manure application performed pre-sowing 2019 at Thuddungra site only
Feb/Mar 2020 SMN versus: | Ardlethan | Girral | Rannock | Temora | Thuddungra |
|---|---|---|---|---|---|
2019 Protein% | 0.17 | 0.49 | 0.59 | 0.51 | 0.67 |
2019 Dry Yield | 0.09 | -0.02 | 0.05 | 0.16 | -0.55 |
2019 N removal | 0.22 | 0.06 | 0.16 | 0.32 | -0.47 |
Elevation | 0.11 | -0.36 | 0.28 | 0.19 | -0.46 |
ECa (0.5) | -0.32 | 0.17 | -0.07 | 0.33 | 0.19 |
ECa (1.0) | -0.34 | 0.19 | 0.19 | 0.41 | 0.24 |
0-60 cm sand% | 0.18 | -0.40 | -0.12 | -0.42 | -0.09 |
0-60 cm clay% | -0.20 | 0.36 | -0.03 | 0.50 | 0.02 |
0-30 cm OC% | 0.23 | 0.47 | 0.44 | 0.32 | 0.39 |
Manure rate* | - | - | - | - | 0.66 |
Importantly, at each of the four significantly correlating sites, areas of the paddock with the lowest protein% coincided reasonably well with areas of low SMN (e.g., see Figure 3 and Figure 4 examples). This occurred both in paddocks of lower overall protein% (Rannock, Temora) and in those with very high protein% levels (Thuddungra, Girral). On the other end of the spectrum, areas within each paddock with the highest protein concentrations did not necessarily always coincide with the highest SMN values (e.g., northern zone in Figure 4). In the two main instances this occurred, these zones were in low lying areas with at least average SMN levels and it is likely they were impacted by frost, which was noted across these sites in 2019. These localised effects demonstrate the importance of paddock and seasonal knowledge when interpreting protein and other data patterns.
When comparing 2019 dry yield and N removal to patterns of 2020 start-of-season SMN there was a very poor relationship (non-significant) at four out of five sites (Figure 2b; Table 3). This result is not unexpected given that moisture supply was by far the most limiting factor to yield in 2019 (not N supply). At the fifth site (Thuddungra), SMN and yield correlated negatively, i.e., higher SMN coincided with lower yields. This is likely explained by a VR chicken manure application which was undertaken just prior to sowing in 2019, which increased the severity of ‘haying off’ where rates were highest. This interpretation is also supported by significant positive correlations found between manure rates and both 2020 start-of-season SMN (Table 3) and 2019 protein% (r = 0.69, P < 0.0001).
OC% consistently had a positive correlation with SMN, however the strength of the relationship was variable and not always significant (Table 3). It is possible that this relationship may have been stronger if soil sampling had been delayed until later in the season (following rainfall), however it is also worth considering that OC% is a bulked measurement of particulate, humus and recalcitrant (char-like) carbon fractionates, which vary in their ability to mineralise N (Baldock et al., 2013). Further research would ideally include the measurement of individual carbon fractionates and/or mineralisable N to better capture/understand the spatial patterns of mineralisation N supply.
Of the soil type proxy layers examined (ECa, sand%, clay%), there were no significant correlations with start-of-season SMN observed across all five sites, however two of the sites (Girral and Temora) had significant negative correlations between 0-60 cm sand% and SMN (i.e., sandier soils had lower SMN; Table 3). Along with Ardlethan, these sites were quite variable in soil type characteristics in comparison to Rannock and Thuddungra, where soil types were more consistent.
Previous management history also appeared to be a key driving factor of N variability for at least three sites, with noticeable differences observed between areas that were previously fenced separately, despite some of these changes being made up to 15 years prior.
These results suggest that in any one paddock there are a great number of variables that may potentially (but not always) influence spatial patterns of SMN. This highlights the difficulty of creating accurate management zones to predict patterns of N supply in the absence of higher resolution data to test assumptions around zone homogeneity.
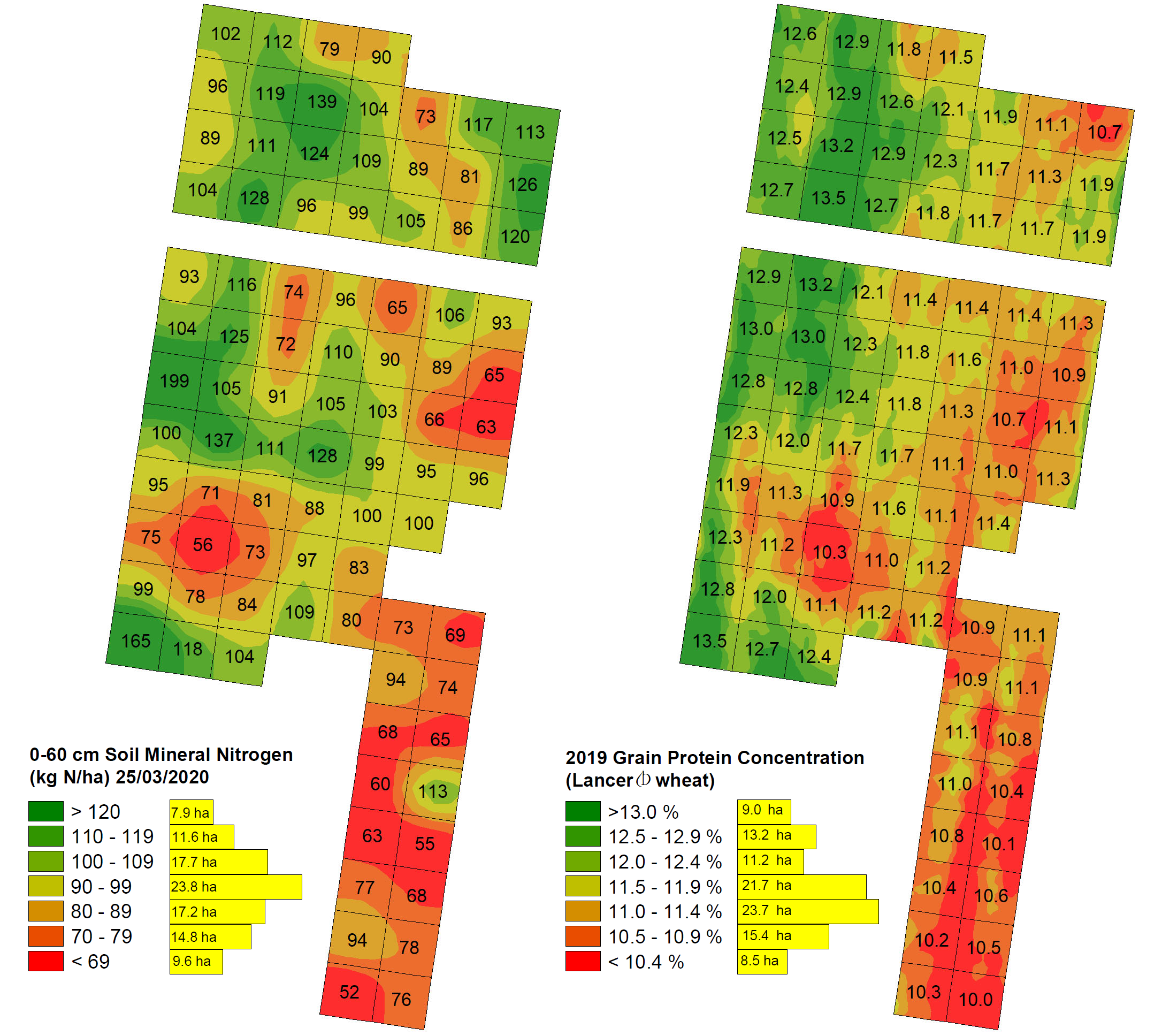
Figure 3. Rannock site 0-60 cm SMN (kg N/ha) sampled 25/03/2020 (left) and 2019 wheat protein% (right). Pearson correlation coefficient (r) = 0.59, P < 0.0001. The missing section between the two blocks is the location of an old fence line which was excluded from the sampling plan. Each cell size is 108 x 108 m, total area = 102.7 ha.
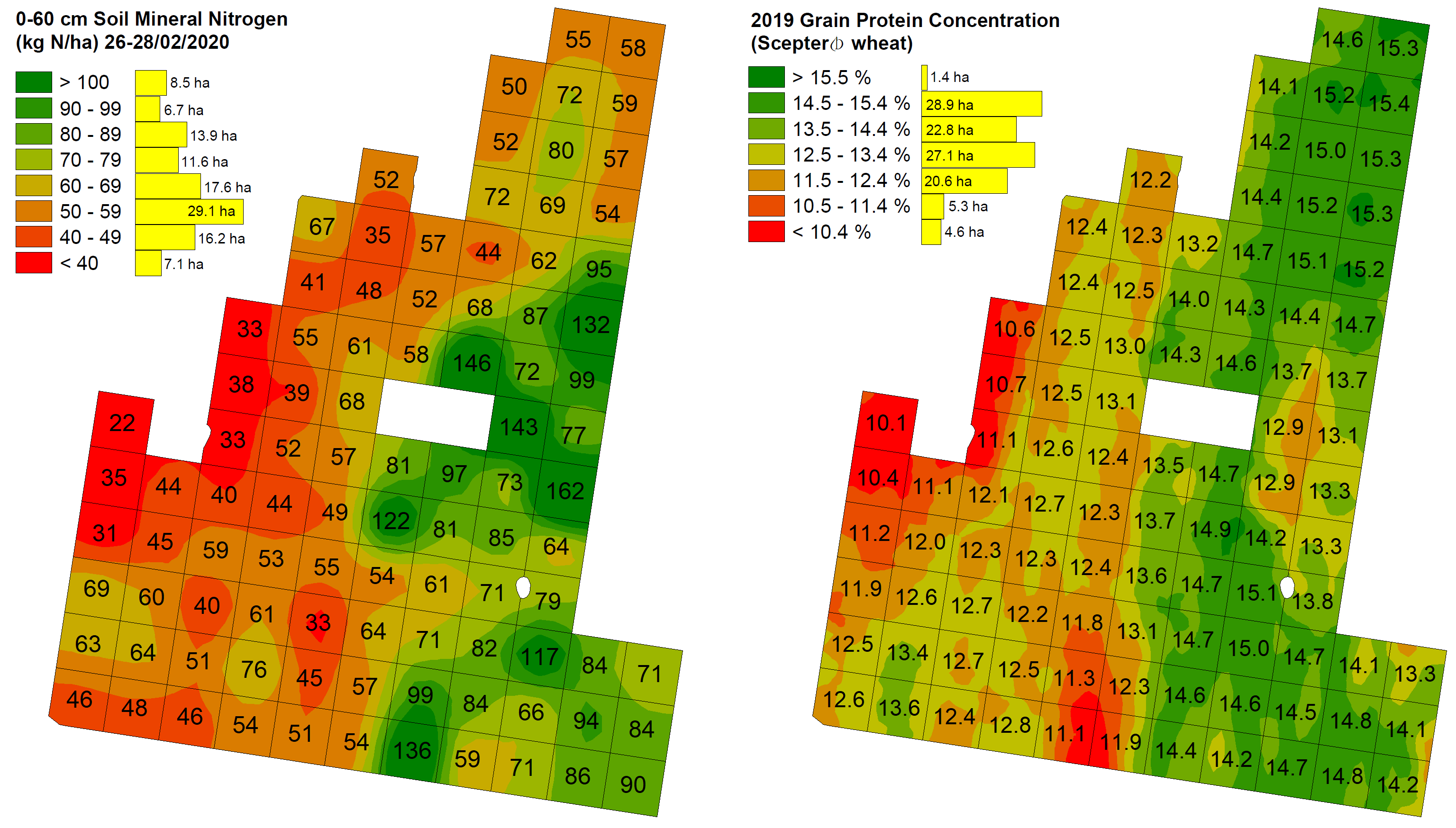
Figure 4. Temora site 0-60 cm SMN (kg N/ha) sampled 26-28/02/2020 (left) and 2019 wheat protein% (right). Pearson correlation coefficient (r) = 0.51, P < 0.0001. The far north of the paddock is a low-lying area that yielded poorly and was most likely severely impacted by both moisture stress and frost in 2019. Each cell size is 108 x 108 m, total area 110.7 ha.
Across the five sites, there was a general trend of increasing strength of correlation between SMN and protein% as the average SMN level increased. This may be explained by considering that N supply levels have to be high in comparison to N demand in order for there to be substantial residual (carryover) SMN. If crop demand is much higher than supply, SMN may be drawn down across the paddock and residual N will be correspondingly low. In this situation, protein% may still vary, as overall N supply may have differed spatially throughout the season. In this situation, it will be necessary to consider if the factors that caused variability in protein% are likely to be present in the following season or if they were a ‘one-off’. For example, differences in mineralisation occurring due to soil type or long-term management practices are likely to reoccur while differences in carryover N patterns from the previous year may or may not reoccur.
This interpretation may explain the observations at the Ardlethan site, where 2019 protein variability was still quite high (6.7 – 11.9%) despite reasonably low 0-60 cm 2020 start-of-season SMN levels across the paddock (average 46 kg N/ha, Table 2). This suggests that N deficiency occurred across most of the paddock in 2019 (drawing SMN to very low levels), however the magnitude of N deficiency varied. When examining the patterns of protein variability, higher protein levels coincided with lighter textured soils on the eastern third of the paddock while lower protein levels coincided with heavier soils on the western two-thirds of the paddock. One possible explanation is that 2019 start-of-season (carryover) SMN levels differed between these two zones. This explanation is supported by a review of 2018 canola yields which were higher on the heavier soil type (i.e., more N removal occurred). A second explanation may be that additional N was accessed on the lighter soil type below 60 cm depth (i.e., below the depth of sampling). This may have occurred if sub 60 cm N reserves were variable OR if the less hostile subsoil conditions (lower CEC/EC/Cl/Na%) on the lighter soil type allowed greater root penetration during the very dry 2019 season.
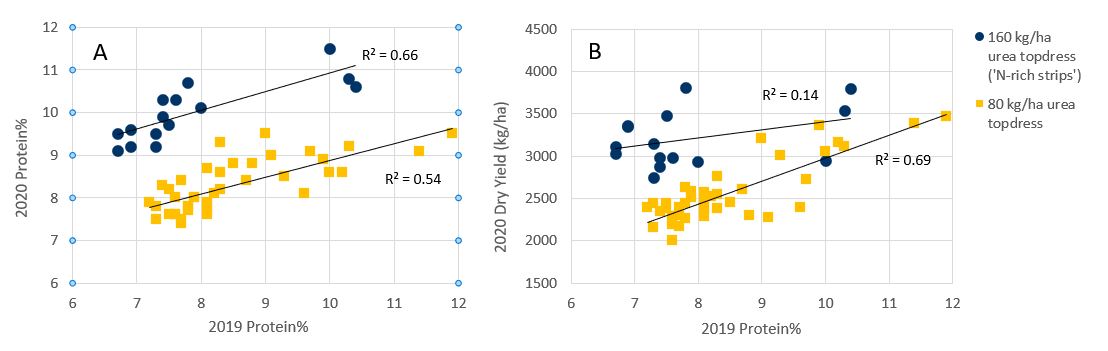
Due to the uncertainty around this result, a strip trial experiment was implemented in 2020 to explore whether the grid soil mapping results or 2019 protein% layer would have been the best basis for site-specific N in 2020. The site was grown to a second season of wheat, with 80 kg/ha urea applied as a flat rate and two 160 kg/ha urea N-rich strips applied at 140 m width.
Results demonstrated a significant positive correlation between 2019 protein% and 2020 protein% for both the N-rich strip areas (n = 15, r = 0.81, P < 0.001) and non N-rich strip areas (n = 40, r = 0.73, P < 0.0001; Figure 5a). A significant positive correlation was also observed between 2019 protein% and 2020 yield for the non N-rich strip areas (r = 0.83, P < 0.0001; Figure 5b) while no significant correlations were observed between 2020 start-of-season SMN and 2020 yield or protein.
Figure 5. 2019 wheat (cv. Lancer) protein% versus 2020 wheat (cv. Spitfire) a) protein% and b) grain yield at the Ardlethan site. Each point represents one 120 x 120 m grid cell. N-rich strips n = 15, non N-rich strips n = 40.
The consistency of protein patterns observed between the 2019 and 2020 seasons despite a lack of correlation with grid soil sampling results suggests there may be either differences in deeper SMN that has not been captured by 0-60 cm soil sampling, differences in the plant accessibility of N present below 60 cm or differences in mineralisation N supply between the two soil zones. The latter explanation appears less likely given that OC% levels were found to be lower on the lighter textured (high protein%) soil zone.
An average yield increase of 564 kg/ha and protein increase of 1.7% was observed for the N-rich strip cells when compared to their immediately adjacent non N-rich cells. A negative relationship was found between 2019 protein% and 2020 yield response, i.e., lower protein% areas had the largest yield response to additional N (r = -0.56, P < 0.01; Figure 5b, Figure 6). There was no significant relationship between yield response and start-of-season SMN (r = -0.04, ns).
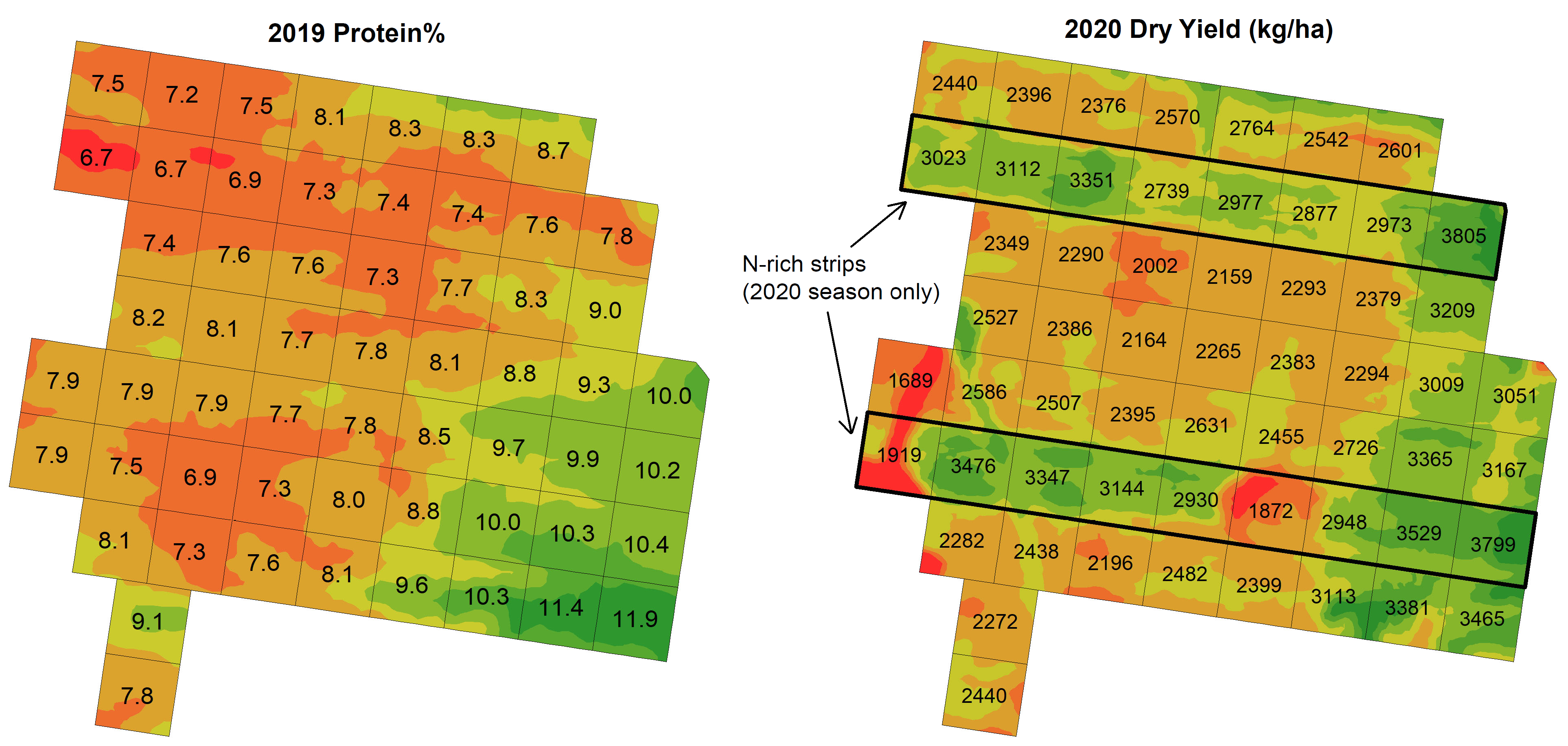
Figure 6. 2019 wheat (cv. Lancer) grain protein (left) and 2020 wheat (cv. Spitfire) dry yield at Ardlethan, with locations of N-rich strips shown. Note the greater yield response to additional N in areas of lower 2019 protein%.
These results suggest that given a fixed N budget, applying additional fertiliser to the lowest protein% areas of the paddock would have produced the greatest overall yield increase. Therefore, it appears that the adoption of a VR N strategy in 2020 based on the 2019 protein% pattern would likely have resulted in a more profitable outcome at this site than using grid soil mapping results or management zones where soil tests were used to directly determine rates.
Conclusions, challenges and further research required
The results of this project and experiences working with growers collecting and utilising harvester protein data have demonstrated that considerable potential exists for protein-based site-specific N strategies to drastically improve N management in our cropping systems, when used in conjunction with annual soil sampling and an appropriate N rate calculation method.
Research results suggested that at higher background SMN levels (where N supply is not drastically exceeded by demand), it is likely that a good relationship will exist between protein% and residual (carryover) N. Even at low (constraining) SMN levels, results showed that protein% patterns may still give a good indication of spatial patterns of N requirements. It is further worth considering that in these instances, considerably higher N rates are probably required in an overall sense, therefore any method that encourages more considered N management (e.g., through reviewing protein levels and undertaking basic ground-truth soil testing) is likely to have a positive impact on profitability.
The success of protein-based site-specific N strategies appears to be linked to the major advantage of this approach whereby the crop itself indicates the N adequacy it experienced over the sum of the whole season. This circumvents many of the challenges of site-specific N management which have either limited the quality/efficacy of some VR N approaches (that attempt to provide simple solutions to a complex problem) or have limited the uptake of other VR N approaches (that are too complex/laborious to be practical). The high spatial resolution of this data and relatively low cost when compared to alternative approaches (e.g., intensive soil sampling) is another major advantage.
While protein maps cannot be used to guide N management decisions in the season of their collection, this method should be considered more of a ‘whole-system’ approach to N management, with the aim of incrementally building (and/or mining) background SMN levels to match site-specific yield potentials across the farming operation over a number of seasons. This approach has considerable synergy with the concept of ‘N banking’ (Hunt et al., 2021; Meier et al., 2021) which aims to decouple N input decisions from seasonal demand by ‘topping up’ N levels each year to a pre-defined target that would be considered non-limiting in most seasons.
By using these two methods in conjunction (on soils that are not prone to losses), growers are armed with a simple, yet targeted strategy to both reduce/eliminate areas of yield loss due to N deficiency and reduce instances of N oversupply which are environmentally, agronomically and economically undesirable. This approach also has logistical benefits in that N rates and VR input maps can be determined/created quite early in the season (following the return of deep N soil test results). This has obvious benefits for financial budgeting and planning however also means that these decisions can be made well ahead of time rather than at a potentially stressful period before a rain event if relying on mid-season remotely sensed imagery, for example.
For this approach to be widely implemented, additional work is required to determine how to bridge the data gap that occurs in seasons where non-cereal crops are grown. Although not discussed in the current paper, results at the four project sites that grew canola in 2020 did not suggest that their protein patterns were as closely related to N supply as those observed in wheat. This may be due to the sensitivity of canola oil/protein concentrations to late seasonal climatic conditions (Walton et al., 1999; Uppal et al., 2019), however additional research is required to further explore this issue (see the full research report for 2020 trial results and a discussion around canola oil/protein drivers - http://www.farmlink.com.au/project/nitrogen-variability).
Another area for further research is examining the impact of frost during grain filling, which can result in elevated grain protein concentrations by curtailing the deposition of starch (Allen et al., 2001). While this may serve to ‘artificially’ elevate grain protein concentrations, it is possible that this effect is counteracted by reduced yields (N removal) and higher N concentrations of residues.
These challenges highlight that the most successful site-specific N management strategies will probably use a number of data layers and grower knowledge in conjunction with protein mapping and targeted deep N sampling to devise effective N input maps over the whole rotation. Such data layers that were identified by the current study to be potentially useful indicators of N variability included soil type parameters (e.g., ECa, CEC or texture mapping, subsoil health tests), long-term productivity (e.g., stacked yield or biomass maps), landscape features (e.g., elevation) and previous management history information (e.g., locations of amalgamated paddocks and their histories).
Given the immense potential productivity and environmental benefits of improved site-specific N management, considerable scope exists for follow up research to address the abovementioned challenges and explore the applicability of these methods in other regions and soil types.
References
Allen, H.M., Pumpa, J.K. and Batten, G.D. (2001) Effect of frost on the quality of samples of Janz wheat. Australian Journal of Experimental Agriculture 41:5, 641-647
Angus, J. F., Bolger, T.P., Kirkegaard, J.A. and Peoples, M.B. (2006) Nitrogen mineralisation in relation to previous crops and pastures. Australian Journal of Soil Research 44:4, 355-365
Angus, J. F. and Grace, P. R. (2017) Nitrogen balance in Australia and nitrogen use efficiency on Australian farms. Soil Research 55:6, 435-450
Baldock, J.A., Hawke, B., Sanderman, J., Macdonald, L.M. (2013) Predicting contents of carbon and its component fractions in Australian soils from diffuse reflectance mid-infrared spectra. Soil Research 51, 577–595.
Basso, B., Fiorentino, C., Cammarano, D. and Schulthess, U. (2016) Variable rate nitrogen fertilizer response in wheat using remote sensing. Precision Agriculture 17, 168-182
Bramley, R.G.V. and Janik, L.J. (2005) Precision Agriculture Demands a New Approach to Soil and Plant Sampling and Analysis—Examples from Australia, Communications in Soil Science and Plant Analysis, 36:1-3, 9-22
Brill, R., Gardner, M., Graham, R. and Fettell, N. (2013) Will low protein become the new norm? GRDC Grower & Advisor Update, Coonabarabran, 25.02.2013. Accessed via https://grdc.com.au/resources-and-publications/grdc-update-papers/tab-content/grdc-update-papers/2013/02/will-low-protein-become-the-new-norm
Colaço, A.F., & Bramley, R.G.V. (2018) Do crop sensors promote improved nitrogen management in grain crops? Field Crops Research, 218, 126-140
Dalal R. C. and Chan K. Y. (2001) Soil organic matter in rainfed cropping systems of the Australian cereal belt. Soil Research 39, 435-464
Fowler, D. B. (2003) Crop Nitrogen Demand and Grain Protein Concentration of Spring and Winter Wheat. Agronomy Journal 95, 260-265
Hochman, Z. and Horan, H. (2018) Causes of wheat yield gaps and opportunities to advance the water-limited yield frontier in Australia. Field Crops Research 228, 20-30
Holford I.C.R., Doyle A.D. and Leckie C.C. (1992) Nitrogen response characteristics of wheat protein in relation to yield responses and their interactions with phosphorus. Australian Journal of Agricultural Research 43, 969-986
Hunt, J., Kirkegaard, J., Maddern, K. and Murray, J. (2021) Strategies for long term management of N across farming systems. GRDC Grower & Advisor Update, Wagga Wagga, 17.02.2021. Accessed via https://grdc.com.au/resources-and-publications/grdc-update-papers/tab-content/grdc-update-papers/2021/02/strategies-for-long-term-management-of-n-across-farming-systems
Lobry de Bruyn, L., & Andrews, S. (2016). Are Australian and United States farmers using soil information for soil health management? Sustainability 8, 1-33
Meier, E.A., Hunt, J.R. and Hochman, Z. (2021) Evaluation of nitrogen bank, a soil nitrogen management strategy for sustainably closing wheat yield gaps. Field Crops Research 261, 108017
Moffitt, E.M., (2021) Utilising new technologies to better manage within-paddock nitrogen variability and sustainably close the yield gap in southern NSW. FarmLink 2020 Research Report. Accessed via: http://www.farmlink.com.au/project/nitrogen-variability
Perry, E.M., Morse-McNabb, E.M., Nuttall, J.G., O’Leary, G.J. and Clark, R. (2014) Managing Wheat From Space: Linking MODIS NDVI and Crop Models for Predicting Australian Dryland Wheat Biomass, IEEE Journal of Selected Topics in Applied Earth Observations and Remote Sensing, 7:9 3724-3731
Rab, M.A., Fisher, P.D., Armstrong, R.D., Abuzar, M., Robinson, N.J., Chandra, S. (2009) Advances in precision agriculture in south-eastern Australia. IV. Spatial variability in plant-available water capacity of soil and its relationship with yield in site-specific management zones. Crop and Pasture Science 60, 885-900
Richetti, J., Colaço, A. and Lawes, R. (2020) Can crop sensors help make nitrogen management decisions? GRDC Grower & Advisor Update, Perth, 11.02.2020. Accessed via https://grdc.com.au/resources-and-publications/grdc-update-papers/tab-content/grdc-update-papers/2020/02/can-crop-sensors-help-make-nitrogen-management-decisions
Russell, J.S. (1963) Nitrogen content of wheat grain as an indication of potential yield response to nitrogen fertilizer. Australian Journal of Experimental Agriculture and Animal Husbandry 3, 319-325
Sandral, G.A., Tavakkoli, E., Harris, F., Koetz, E. (2018) Improving nitrogen fertiliser use efficiency in wheat using mid-row banding. GRDC Grower & Advisor Update, Wagga Wagga, 13.02.2018. Accessed via https://grdc.com.au/resources-and-publications/grdc-update-papers/tab-content/grdc-update-papers/2018/02/improving-nitrogen-fertiliser-use-efficiency-in-wheat-using-mid-row-banding
Schwenke, G., Beange, L., Cameron, J., Bell, M., Harden, S. (2019) What soil information do crop advisors use to develop nitrogen fertilizer recommendations for grain growers in New South Wales, Australia? Soil Use and Management, 35, 85-93
Simmonds, N.W. (1995), The relation between yield and protein in cereal grain. Journal of the Science of Food and Agriculture, 67: 309-315.
Unkovich M.J., Herridge D.F., Denton M.D., McDonald G.K., McNeill A.M., Long W, Farquharson, R. and Malcolm, B. (2020) A nitrogen reference manual for the southern cropping region. GRDC publication. Accessed via https://grdc.com.au/resources-and-publications/all-publications/publications/2020/a-nitrogen-reference-manual-for-the-southern-cropping-region
Uppal, R.K, Brill, R. and Bromfield, J. (2019) Effect of heat stress on canola grain yield and quality. GRDC Grower & Advisor Update, Wagga Wagga, 19.02.2019. Accessed via https://grdc.com.au/resources-and-publications/grdc-update-papers/tab-content/grdc-update-papers/2019/02/effect-of-heat-stress-on-canola-grain-yield-and-quality
Walton, G., Si, P. and Bowden, B. (1999) Environmental Impact on Canola Yield and Oil. In: ‘Proceedings of the 10th International Rapeseed Congress’. Canberra, ACT. (Agriculture Western Australia: South Perth, W. Aust.) Accessed via http://www.regional.org.au/au/gcirc/2/136.htm
Acknowledgements
This research component of this paper was undertaken in partnership between FarmLink Research and Precision Agriculture. It was supported by the Department of Agriculture, Water and the Environment through funding from the Australian Government’s National Landcare Program.Research was further supported by Charles Sturt University where the author holds an Adjunct Research Fellow position.
A very big thank you to the five participating growers for providing your paddocks, time and input to the project. Thanks to Dr Kirsten Barlow (Precision Agriculture) for assistance in data analysis, the many discussions and review of papers. Thanks also to Dr John Angus (CSIRO) for discussions around N budgeting methods and Mat Clancy (Next Instruments) for technical support. The support of APAL agricultural laboratory who provided MIR Particle Size Analysis in-kind is appreciated.
Contact details
Eva Moffitt
EM Ag Consulting
Barmedman, NSW 2668
Ph: 0437 189025
Email: eva.moffitt@outlook.com
Varieties displaying this symbol beside them are protected under the Plant Breeders Rights Act 1994
® Registered trademark
Was this page helpful?
YOUR FEEDBACK