Update on a dedicated sampling strategy to improve the accuracy of PreDicta B soil testing
Author: Steven Simpfendorfer (NSW DPI, Tamworth) Alan McKay and Shawn Rowe (SARDI, Adelaide) | Date: 03 Aug 2015
Take home messages
- PreDicta B® is a good technique for identifying the level of risk for crown rot (and other soil-borne pathogens) prior to sowing within paddocks. However, this requires a dedicated sampling strategy and IS NOT a simple add on to a soil nutrition test.
- Soil cores should be targeted at the previous winter cereal row if evident and RETAIN any stubble fragments.
- Short pieces of stubble (two from each PreDicta B® soil sampling location) from previous winter cereal crops and/or grass weed residues can be added to the soil sample to enhance detection of the Fusarium spp. that cause crown rot.
- ‘Spiking’ with stubble will reduce the likelihood of ‘failure to warn’ situations for crown rot but unfortunately will also increase the probability of false warnings.
Introduction
PreDicta B® is a DNA based soil test which detects levels of a range of cereal pathogens that is commercially available to growers through the South Australian Research and Development Institute (SARDI). The main pathogens of interest in the northern grains region detected by PreDicta B® are Fusarium spp. (crown rot), Bipolaris sorokiniana (common root rot), Pythium (damping off) and both Pratylenchus thornei and P. neglectus (root lesion nematodes, RLNs). Over recent years PreDicta B® has been shown to be a reliable method for assessing RLN populations but is perceived by industry to be less reliable in assessing levels of crown rot risk in the northern region.
Between 2010 and 2012, we conducted an annual winter cereal pathogen survey of 248 paddocks across 12 districts in central and northern NSW. The three-year survey measured the DNA levels of the Fusarium pathogen at sowing against the infection levels that had developed by harvest. This research found that in 75% of paddocks, PreDicta B® at sowing predicted the actual level of infection that developed in the crop as measured after harvest within one risk category. In 3% of paddocks PreDicta B® overestimated the risk of infection compared to actual development levels (false warning) but of more concern was that PreDicta B® underestimated the risk of crown rot in 22% of paddocks (failure to warn).
The underestimation of crown rot risk is potentially due to the crown rot fungus being stubble-borne while PreDicta B® is a soil based test. Further investigation found that soil nutrition sampling strategies were often being used to collect both the soil nutrition and PreDicta B® samples. This is significant because soil nutrition samples are normally collected between the rows with stubble removed whereas PreDicta B® samples need to be collected along the row of the previous cereal crop and incorporate any stubble residues.
Improving the accuracy and calibrating PreDicta B® in the northern region for crown rot is important for advisers and growers to better plan their crop and varietal selection prior to winter crop sowing and avoid costly yield losses from this disease. This is particularly relevant leading into the 2015 cropping season with a high durum grain price last season likely to see many growers considering durum this year. Durum is highly susceptible to crown rot so the cost of getting it wrong and sowing into a high disease risk paddock is significant.
The following paper reports on collaborative research conducted by NSW DPI and SARDI across central/northern NSW in 2013 to improve the accuracy of the PreDicta B® test in assessing crown rot risk by fine tuning soil sampling techniques and recommendations.
Detection issue?
Currently there are three separate tests within PreDicta B® that detect common Fusarium species causing crown rot across Australia - two tests which detect variations in F. pseudograminearum populations and a third test which detects both F. culmorum and F. graminearum but cannot differentiate between these two species. The failure to warn of the risk of crown rot in 22% of paddocks could be related to the inability of the current PreDicta B® tests to actually detect other species/variants of Fusarium causing crown rot across the region. A national survey was conducted in 2013 and 2014 with over 800 Fusarium isolates collected from wheat and barley plants with basal browning characteristic of crown rot infection from across Australia. Molecular analysis determined that all Fusarium species known to cause crown rot are being detected by the current PreDicta B® assays. Hence, there is no detection issue with the current PreDicta B® tests that could contribute to the underestimation of crown rot risk.
Does the addition of stubble (‘spiking’) improve PreDicta B® assessment of crown rot risk?
In 2013 each of the six ranges in 13 cereal NVT sites and 8 NSW DPI district pathology (DP) trials were cored using PreDicta B® (Table 1). Two separate soil samples were collected from each range at each of the 21 field sites spread from central NSW up into southern Qld. All cores were targeted at the previous winter cereal rows if evident. Previous winter cereal crop stubble was also collected across each separate range at coring if present and used to spike set soil samples. Twenty-five lowest nodes (1 cm segments around node) were cut from the corresponding stubble sample and added to one of the samples collected from each range. All samples were then sent to SARDI for PreDicta B® analysis.
Table 1. Location of field trial sites in 2013
|
Site no. |
Location |
Site No. |
Location |
|
1 |
NVT Bellata |
12 |
NVT Westmar |
|
2 |
NVT Bullarah |
13 |
NVT Wongarbon |
|
3 |
NVT Coolah |
14 |
DP Narrabri |
|
4 |
NVT Coonamble |
15 |
DP Terry Hie Hie |
|
5 |
NVT Gilgandra |
16 |
DP Bithramere |
|
6 |
NVT Macalister |
17 |
DP Spring Ridge |
|
7 |
NVT Merriwa |
18 |
DP Tamworth |
|
8 |
NVT North Star |
19 |
DP Garah |
|
9 |
NVT Spring Ridge |
20 |
DP Rowena |
|
10 |
NVT Trangie |
21 |
DP Macalister |
|
11 |
NVT Tulloona |
||
After harvest stubble was collected from all plots of three varieties (EGA Gregory, Suntop and Caparoi or Spitfire) at each site. Twenty-five crowns from each plot were trimmed, surface sterilised and plated onto laboratory media to determine the incidence of crown rot infection that developed during the 2013 based on the recover of Fusarium.
PreDicta B® risk for crown rot is a sum of all three Fusarium tests which are then converted to a log scale to normalise the data. Current PreDicta B® crown rot risk categories for durum wheat are used in the northern region and corresponding harvest infection levels based on plating have also been developed for the region (Table 2).
Table 2. Current PreDicta B® crown rot risk levels and corresponding harvest infection level
|
PreDicta B® |
Risk or harvest disease level |
Incidence of infection |
|
<0.6 |
Below detection limit (BDL) |
<2 |
|---|---|---|
|
0.6 – 1.4 |
Low |
3-12 |
|
1.4 – 2.0 |
Medium |
13-24 |
|
>2.0 |
High |
≥25 |
Addition of stubble fragments to soil samples was only possible at few sites with previous cereal stubble being present at 7 (red larger diamonds, Figures 1 & 2) of the 21 sites (2, 4, 5, 8, 12, 16 and 18). The addition of stubble increased the crown rot risk level at six of the sites, from low to high at 2 sites (sites 4, NVT Coonamble and 12, NVT Westmar), low to medium at 2 sites (sites 5, NVT Gilgandra and 8, NVT North Star) and medium to high at 2 sites (sites 2, NVT Bullarah and 18, DP Tamworth)(Figure 1 and 2). For the last site (site 16, DP Bithramere) the log Fusarium DNA/g increased from 2.5 to 4.3 with the addition of stubble but this did not increase the predicted crown rot risk level as both values represented a high risk of crown rot development.
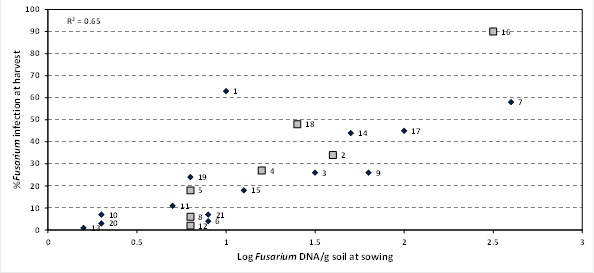
Figure 1. Relationship between at sowing DNA levels of Fusarium using PreDicta B® and incidence of crown rot infection at harvest – ‘Unspiked’ samples in 2013
Sites spiked with stubble represented by larger grey squares (sites 2, 4, 5, 8, 12, 16 and 18)
There was a 65% correlation between unspiked PreDicta B® results collected at sowing and the actual incidence of crown rot infection that developed by harvest (Figure 1). Fourteen sites in 2013 had a low or BDL risk for crown rot development based on unspiked PreDicta B® soil tests at sowing. At 8 of these sites (6, 8, 10, 11, 12, 13, 20 and 21) the DNA test correctly predicted the actual level of disease which developed while at 6 sites (1, 4, 5, 15, 18 and 19) PreDicta B® underestimated the risk of disease development. This is generally considered a ‘failure to warn’ and was particularly evident at site 1 (NVT Bellata) where only a 1.0 log Fusarium DNA value was measured at sowing but 63% of plants were infected with crown rot at harvest. In the medium risk category the DNA test correctly predicted the incidence of disease development at two sites (3 and 9) but underestimated the risk at two sites (2 and 14). All three sites predicted to be in the high risk category by the DNA test at sowing (sites 7, 16 and 17) did develop high infection levels during the 2013 season (Figure 1).
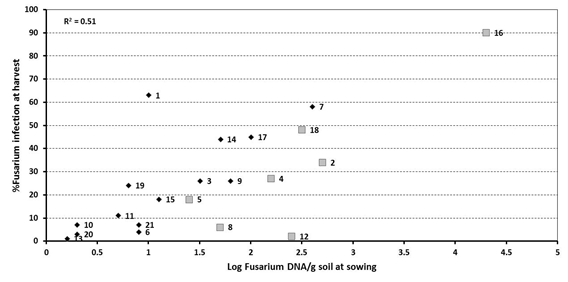
Figure 2. Effect of ‘spiked’ samples at 7 sites on the relationship between at sowing DNA levels of Fusarium using PreDicta B® and incidence of crown rot infection at harvest – 2013.
Sites spiked with stubble represented by larger grey squares (sites 2, 4, 5, 8, 12, 16 and 18)
The addition of stubble to the PreDicta B® soil samples (‘spiking’) collected from seven of the 21 sites at sowing reduced the correlation with the incidence of plants infected with crown rot at harvest down to 51% (Figure 2). However, spiking with stubble at sites 4, 5 and 18 removed them from the low risk category into their correct level of disease incidence at harvest. That is, these were no longer ‘failure to warn’ situations, with the stubble spiking correctly predicting the risk at sowing of what developed by harvest in the crop. Stubble spiking also corrected the underestimation of risk from medium to high at site 2. Spiking did not change risk categories at site 16 which was high with both the unspiked and spiked soil samples. However, the higher DNA level in the spiked sample better reflected the higher disease incidence (90%) at this site relative to other sites (max. 63%) in 2013.
Unfortunately, in two situations (sites 8 and 12) spiking led to an overestimation of the crown rot risk at sowing. Spiking pushed site 8 into a medium risk category but only 6% of plants were infected at harvest while site 12 was pushed into a high risk category with only 2% of plants infected at harvest. These situations would be considered false positives and potentially lead to a missed opportunity for growers where they could have grown a winter cereal crop with minimal risk of yield loss from crown rot.
Conclusions
PreDicta B® is a soil based test so with the collection of cores targeted at the previous winter cereal rows it can provide a good measure of Fusarium levels in the crowns below ground but is restricted in its ability to detect levels in above ground stubble. Adding stubble (‘spiking’) is likely to increase the overestimation of crown rot risk (false positives) while reducing the likelihood of underestimation or ‘failure to warn’ which we consider a preferred situation for growers.
The addition of stubble is also likely to reduce sampling issues following wetter summers (e.g. some northern regions in 2014/15) which can result in greater survival of Fusarium in above ground residues. Significant summer rainfall can lead to rapid decomposition of the crowns of previous cereal crops below ground which reduces the survival of Fusarium in this tissue. However, standing stubble dries out relatively quickly following rainfall events and can hence harbour crown rot inoculum for an extended period. Soil sampling, even targeted at the previous cereal row, will only detect Fusarium levels in the crowns. Hence, the addition of stubble to PreDicta B® soil tests will compensate for situations where there is still significant survival of Fusarium in above ground residues.
Recent collaborative research in the northern region between SARDI and NSW DPI has demonstrated that use of a smaller diameter (1 cm) soil core (e.g. Accucore) to collect 30-45 cores (depending on sampling depth) targeted at the previous cereal row if evident provides a good measure of both RLN and crown rot risk along with a range of other pathogens. This number of cores collected spatially across the paddock is required to account for the potential variability in the distribution of crown rot inoculum.
This research was continued across further sites in the northern region in 2014 and expanded to around 160 NVT sites nationally. This will facilitate further refinement of sampling strategies and calibrate risk categories across regions.
Recommended PreDicta B® sampling strategy for crown rot
These findings have resulted in amended sampling strategy recommendations to improve the value of PreDicta B® as a management tool for crown rot. To correctly sample, growers and advisors should:
- Collect three cores of 1 cm diameter and 15 cm deep from each of 15 different locations within the target paddock or production zone. Samples may be taken to 30 cm depth in the northern region if concerned about Pratylenchus thornei detection. If using a larger diameter core or coring to 30 cm, take fewer cores per location.
- Take the soil cores from along/in the rows of previous cereal crop if still visible and retain any stubble collected by the core (most soil borne pathogens are concentrated under the rows of the last cereal). Sampling depth (0-15 cm or 0-30 cm) does not appear to greatly impact on detection of the various pathogen levels in the northern region when the collection of cores is targeted at the previous cereal rows. However, the actual sampling depth needs to be recorded on the sample bag when collected as it is used to refine reporting of results to adjust for pathogens which are more concentrated at the soil surface.
- If the rows cannot be seen, take the cores at random.
- Add two piece of cereal stubble (if present) to the sample bag at each of the 15 sampling locations to improve the detection of crown rot. Each piece should be a single dominant tiller from the base of different plants and include the crown to the first node (discard material from above the first node).
- The maximum sample weight should not exceed 500 g.
- Significant stubble disturbance such as through harrowing, cultivation or mulching increases the risk of crown rot development if the stubble is infected with Fusarium and collection of soil samples prior to stubble disturbance is likely to underestimate the crown rot risk.
Acknowledgments
This project was co-funded by NSW DPI, SARDI and GRDC under the national improved molecular diagnostics for disease management project (DAS00137) and the national crown rot epidemiology and management program (DAN00175). Assistance provided by Robyn Shapland, Finn Fensbo, Patrick Mortell, Kay Warren, Tara Burns and Karen Cassin (NSW DPI); Herdina, Russell Burns, Aidan Thomson, Ina Dumitrescu, Danuta Pounsett, Irena Dadej, Daniele Giblot-Ducray (SARDI); and Diane Hartley (CSIRO) is greatly appreciated.
Contact details
Dr Steven Simpfendorfer
NSW DPI
0439 581 672
steven.simpfendorfer@dpi.nsw.gov.au
Dr Alan McKay
SARDI
(08) 8303 9375
alan.mckay@sa.gov.au
GRDC Project Code: DAS00137; DAN00175,
Was this page helpful?
YOUR FEEDBACK
