From the lab to the field the scale and impact of fungicide resistance in Australia
Author: F. Lopez-Ruiz, A. Kay, M. Tucker, W. Mair, W. Deng, S. Chang, K. Dodhia, B. Cox, L. Harper and R. Oliver (Centre for Crop Disease Management, Department of Environment and Agriculture, Curtin University) | Date: 23 Feb 2016
Understanding the development of fungicide resistance in Australia
Broadacre cropping in Australia is characterised by low and intermittent rainfall and low levels of soil fertility; average yields are low by international standards. For these reasons the widespread adoption of the use of multiple fungicide treatments did not begin in earnest until about 12 years ago. Many first generation fungicides patents had expired so the products were cheap enough to become economically rational options for disease control. One consequence was that a small number of actives from a single mode of action group – the DMIs or triazoles – dominated the market.
The triazoles remain the backbone of crop protection in broad-acre cropping worldwide and no catastrophic cases of resistance have been reported anywhere in the world. For these reasons, the monitoring of fungicide resistance in Australian agriculture generally was considered unimportant until comparatively recently. The GRDC and later the GWRDC (now AGWA) began funding fungicide resistance work with our group in 2009. Today we can see that the situation in Australia is at a crossroad, with significant levels of resistance in all studied crops.
So far nine cases of resistance have been detected in the last three years (Table 1); many horticultural crops have not been studied but anecdotal evidence suggests that many more cases remain to be uncovered. A few important key factors that currently contribute to this problem are:
1. The host abundance
In the Australian grain belt, large areas of land are devoted to the same crops and varieties. Thus, pathogens do not struggle to find a host before, during and after the growing season.
a. Not having enough break points in the system allows for a constant carryover of disease from one season to the next making it easier for a newly developed fungicide resistant or more virulent strain (i.e. fungicide resistant barley powdery mildew or the new leaf rust pathotype detected in 2015 in Western Australia) to thrive and become established in the field. This situation also gives the newly evolved strains the environment to compete with the often fitter currently established natural populations of the disease which otherwise would out-compete the new strains under conventional paddock management.
b. At the same time, this particularity of the Australian agrosystem creates artificial disease highways that communicate distant areas that otherwise would be isolated. Under these circumstances, any new fungicide resistance cases (and virulence issues) can spread quickly across the country as oppose to a different system where the crops are more fragmented and issues are contained and dealt with more efficiently.
Table 1: Fungicide resistance cases identified in Australia during the period 2014 – 2015.
| Disease | Fungicide |
|---|---|
| Barley powdery mildewb | DMI |
| Wheat powdery mildewa | DMI |
| Canola blackleg | MAP/Kinase |
| Legume Botrytisb | MBC |
| Barley net-blotchesb | DMI |
| Wheat septoria leaf blotchc | DMI |
| Grape powdery mildew b | Qol/DMI |
| Grape downy mildewc | PAA |
| Grape Botrytisb | MBC/E1/E3/D1 |
| aIdentified by the Fungicide Resistance Group (FRG) in 2015 bIdentified by the FRG 2012-2014 cIdentified by other researchers |
|
2. The varietal choice and the market trend
The use of susceptible varieties speeds up the development of resistance. Despite the existence of varieties with good levels of resistance against most diseases, sensitive varieties are sown in many cases and, despite the availability of some good fungicides, poor levels of chemical protection are provided. Again, this is a collateral effect of current market demands and low margins, and leaves growers with very little room to deploy adequate anti-resistance management strategies in their paddocks.
a. Varietal resistance and fungicides are a tandem team. If one is not there to protect the other, or underperforms, then pathogens find it easier to develop virulence to the first or resistance to the latter. Thus, having the right tandem team is central for a successful performance and long-lasting disease management strategy in the field.
b. The typical and probably most discussed example is the historical tandem made by the use of barley varieties, very sensitive to powdery mildew, together with old and cheap fungicides (figure 1). Although barley varieties based on the mlo gene resistant to the disease have been used successfully for many years overseas, there is a general reticence to introduce this type of resistance in the Australian barley varietal pool due to suspected lower yield and increased susceptibility to other diseases such as necrotrophic pathogens.
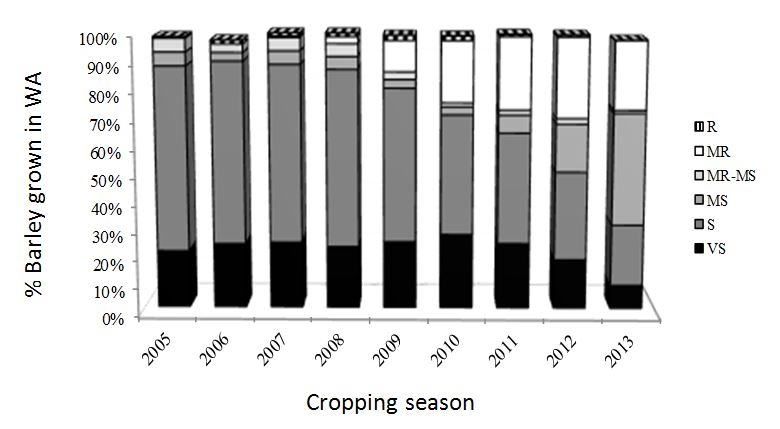
Figure 1: Summary of barley varieties grown in Western Australia during the period 2005–2013. Varieties are classified as resistant (R), moderately resistant (MR), moderately resistant-moderately susceptible (MR-MS), moderately susceptible (MS), susceptible (S) and very susceptible (VS). Source: Barley Variety Guide DAFWA, Bulletin 4826.
3. The arsenal
Until recently, Australian growers were left to manage diseases with old, cheap and often, not very efficient chemistry. A lack of modes of action (MOA) and a very poor stewardship system have been a burden that will require a few years to mitigate. This situation is evident when the number of MOA available for the management of barley powdery mildew in UK (17) is compared with those available in Australia for the same disease (3), or simply by asking growers how often did they spray the same active during the last five seasons. Just as in modern warfare armies need new weapons to defeat an increasingly intelligent enemy, growers need new MOAs that allow them to fight and defeat diseases evolving new fungicide resistances with the guarantee that they are not creating a ‘snowball’ issue due to the lack of adequate stewardship and monitoring resources.
4. The extension component
To our knowledge, efforts made to communicate key messages in the area of fungicide resistance mitigation and management have been quite ‘mild’ to date. The GRDC recently funded a project (CUR00022) aimed at filling the existing gap in knowledge available for growers and advisers by distilling fungicide resistance management information from the research that is carried out both in Australia and overseas. The industry needs an integrated extension program aimed at disseminating the most important anti-resistance strategies and recommendations in order to be successful at identifying and dealing efficiently with the next case of fungicide resistance in the field.
The delay in the wide scale use of fungicides gives Australia the opportunity to learn from the mistakes made in other countries. However, the sad story is that much of the same mistakes have been made:
- Fungicide resistance in barley powdery mildew was known as early as in 1981, yet for ten years barley growers have been using DMI fungicides almost exclusively to deal with the disease. This has led to the development of the biggest and most expensive fungicide resistance epidemic ever registered in the world. It does not help the fact that this situation could have been avoided by looking at what other countries did in the past. However, it is important to keep this wound open to remind the industry what the right path to follow is in order to develop a sustainable agriculture and avoid similar scenarios in the future.
- Products are developed targeting different agrosystem settings overseas where margins generally allow several sprays with different MOAs. As a result, stewardship strategies are developed following the same principle. This is a counterproductive model if these same products are then transferred to a system (Australia) where conditions such as weather, soil, margins and disease pressure, to name a few, differ greatly from those originally used in their development. Under these circumstances fungicides are exposed to a much higher pressure with a greater possibility of resistance developing and becoming established in the population. There is not much we can do about the products themselves with the exception of choosing only those actives able to best perform under harsh Australian conditions. However, the stewardship of the active should be completely reconsidered in view of the existing differences and adapted to take into account key elements such as the limitation in the number of sprays due to current margins in most crops, which impacts the development of anti-resistance management strategies including multiple MOAs.
- Fungicide resistance monitoring strategies have been designed to tackle disease problems at a scale that differs from the Australian agrosystems. Currently, Australia’s land devoted to wheat production is almost as big as the land dedicated to wheat crops in the whole Western Europe. However, varietal choice, climatic conditions, geographical fragmentation and country-specific chemical management, makes both systems hardly comparable. Thus, monitoring strategies developed for such a heterogeneous area cannot respond to Australian grower’s needs for faster turnaround times and knowledge on new resistance mechanisms.
Using the disease’s weaknesses
An adequate knowledge of the enemy confers you a tactical advantage that sometimes is sufficient to win the battle. A grower can make use of the knowledge available on specific diseases in order to take advantage of potential weak points in their biology (i.e. life cycle, host range, etc.) that can be exploited as part of a disease management strategy.
The same principle applies to fungicide resistance. Often, strains showing resistance to fungicides are less fit than the wild type populations due to the negative effect that mutations conferring resistance have in the overall fitness of the strain. This means that these resistant strains would only be competitive while fungicide pressure is maintained. However, if fungicide pressure is removed (stop spraying or change of MOA) wild type populations should outcompete resistant populations and colonize the system again. This is the theory but unfortunately for growers the practical side of it is more complex. What happens in reality is that resistant populations are sometimes almost as fit as the wild type ones. At the same time it is extremely difficult to stop fungicide usage from a specific MOA in an area big enough to impact the resistant population, and use of another fungicide only makes sense if it is from a different MOA. However, such different MOAs are expensive and thus rarely used.
However, sometimes researchers are lucky enough to discover hidden mechanisms buried deep in the DNA of the pathogens that can help growers fight some diseases. This is the case of the negative cross-resistance phenomenon in triazole resistant barley powdery mildew. Using a different fungicide to control an epidemic of a fungus resistant to a given fungicide works because both the resistant and susceptible strains are equally sensitive to the second fungicide. What if, however, the strain resistant to the first fungicide was especially sensitive to the second fungicide? In this case, the second fungicide has a bigger impact on the resistant than the sensitive strain. This favours the sensitive strains and restores sensitivity in the population to the first (and likely cheaper) fungicide.
By analysing the role of each mutation found in the resistant phenotype, researchers have been able to find a case were resistant mutant strains are more sensitive to the triazole fluquinconazole than the wild types (Table 2). In order to achieve this, researchers had to investigate the impact of each mutation on the molecular structure of the triazole target, named CYP51, to find that there was a mutation that made the triazole target bind easier to fluquinconazole but not to other triazoles. The analysis of the conformational changes of the triazole target in resistant isolates carrying this mutation allowed researchers to understand the mechanism behind this phenomenon.
Table 2: Fungicide sensitivity measured as Effective Concentration inhibiting 50 per cent of the growth (EC50) in wild type and resistant barley powdery mildew isolates against several azoles.| Triazole | EC50 (mgL-1) | |
|---|---|---|
| Wild type | Resistant | |
| Tebuconazole | 1.7 | 29.8 |
| Epoxiconazole | 0.6 | 2.6 |
| Propiconazole | 1 | 3.7 |
| Triadimefon | 0.5 | 1.8 |
| Prothioconazole | 0.5 | 6 |
| Flutriafol | 0.3 | 4 |
| Cyproconazole | 0.9 | 2.4 |
| Fluquinconazole | 7.8 | 5.2 |
The need to tailor our management
Thus, the two key questions that open up now are:
- Is negative cross-resistance a widespread phenomenon in agriculture? If so, is it present in most grain diseases?
- Would these findings allow us to design chemical treatments in which different fungicides (DMIs in this case) are sprayed depending on the presence of resistant and/or wild type isolates?
The answer to the first question is quite straightforward. We do not know yet as more research needs to be done. However, in those cases where resistance is due to mutations in the fungicide target site, it is conceivable that some molecules in a MOA group will be more effective than others. Researchers will need to determine not only if this type of molecules exist, but also if the molecules offer an advantage compared with other available fungicides from different MOA groups (i.e. cheaper treatment) and meet the requirements for use under harsh Australian conditions.
The answer to the second question is guided by common sense. The same as we take different antibiotics from the same chemical group for the treatment of different diseases, or even for more virulent versions of the same disease, tailored use of fungicides is required based on the specific combination of disease genotypes found in a paddock. In order to deal with fungicide resistance more efficiently, this needs to become a reality in the near future. A key milestone down this pathway has been the recent development of high throughput monitoring methodologies that allows for the large scale analysis of bulk samples (i.e. a few dozen samples from the same paddock) with really fast turnaround times (24-48h) and at an affordable price ($15-20). Faster turnaround times and tailored treatments are in line with a more efficient and cost-effective way of managing diseases. A grower needs to know if there are better products available for the management of specific disease strains in a timely manner. This resource will improve our current approach to managing resistance by only using those fungicides that are highly effective against the strains detected in the field, saving the grower time and money.
Conclusion
Many growers will experience a disappointing result with a fungicide spray at some stage. This could be due to application problems — e.g. the conditions were wrong for spraying — or the product was not appropriate for the diseases. Or it could be due to the build-up of resistance in the disease population. The key issue is; how do growers respond to such a situation of failed disease control? Spraying again with a different fungicide may control the disease or it may make the resistance problem worse, especially if the second fungicide application is the same MOA or even, the same active. Understanding how fungicide resistance occurs will enable us to minimise the impact and delay the worst effects
The GRDC is currently funding several fungicide resistance research projects in Australia with three main goals:
- Understanding the current status of the issue in grain crops
- Developing tools for the analysis and monitoring of resistance
- Designing better anti-resistance management strategies.
A profound knowledge of the molecular mechanisms by which fungi becomes resistant to fungicides is key to developing new, more efficient and, most importantly, cheaper ways to control and mitigate the build-up of fungicide resistant populations in the field.
N. B. Please note that reference to a fungicide in this paper does not constitute a recommendation for control of a specific disease.
Acknowledgements
Funding for this work was provided through the GRDC Projects CUR00016, CUR00019, CUR00022, DAW00229 and their support is gratefully acknowledged.
Contact details
Fran LopezKent Street, Building 303, Bentley, 6845, Western Australia
08 9266 3061
fran.lopezruiz@curtin.edu.au
Was this page helpful?
YOUR FEEDBACK
